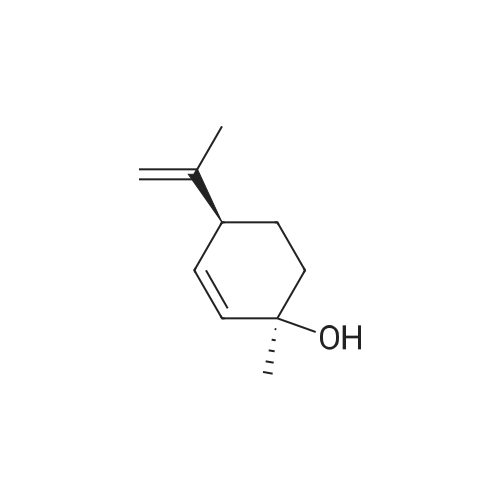
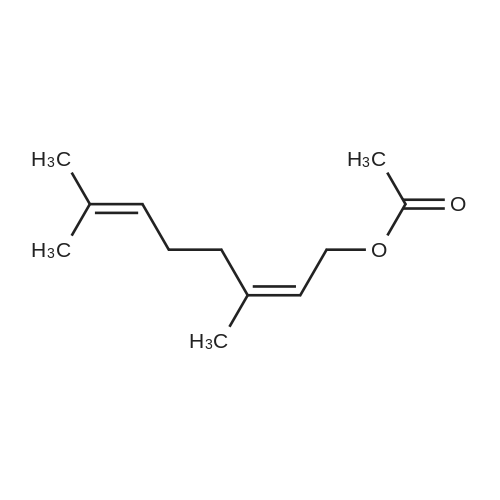
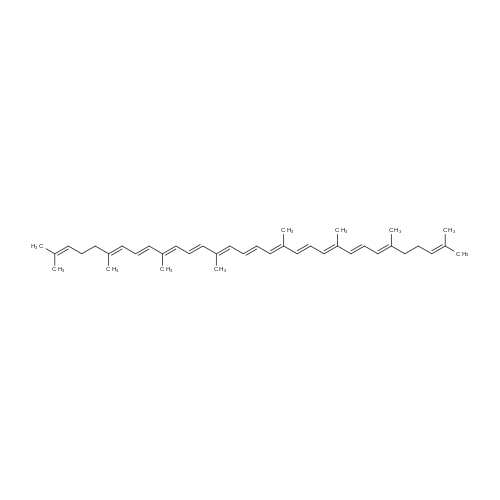
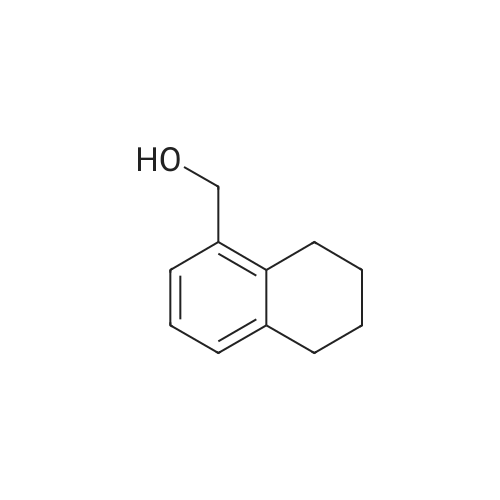
| 95% |
With titanium(IV) isopropylate; tert.-butylhydroperoxide; diethyl (2R,3R)-tartrate; In decane; dichloromethane; acetonitrile; at -40℃; for 10h;Inert atmosphere; Molecular sieve; Cooling with ice; |
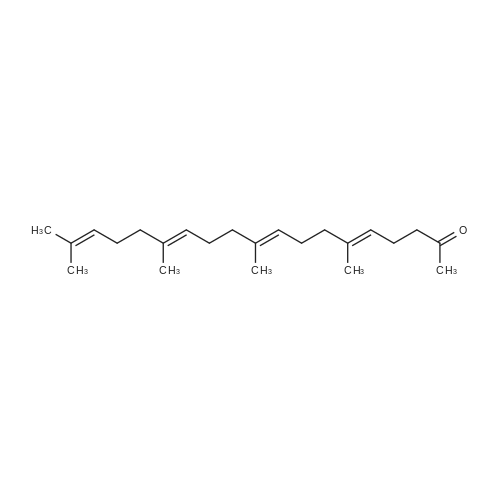
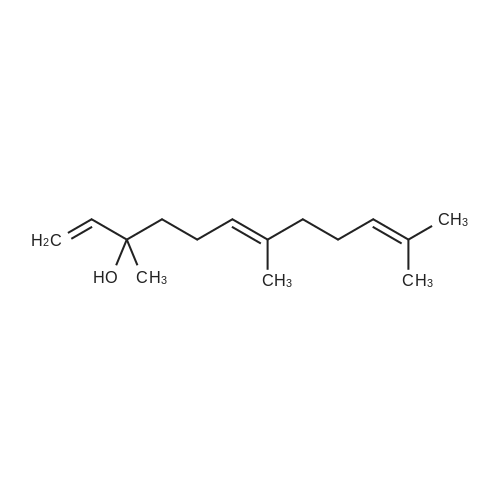
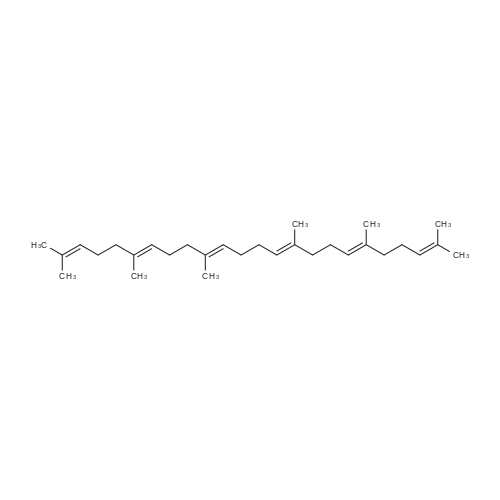
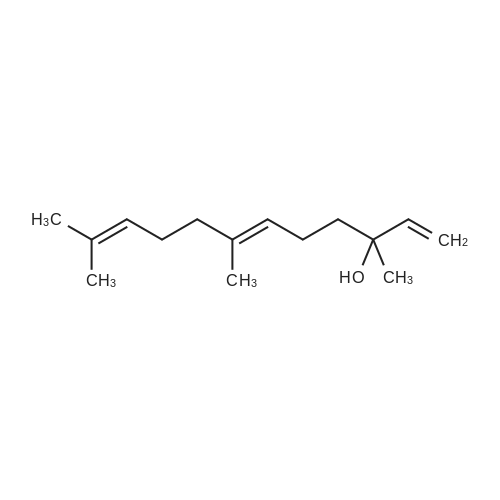
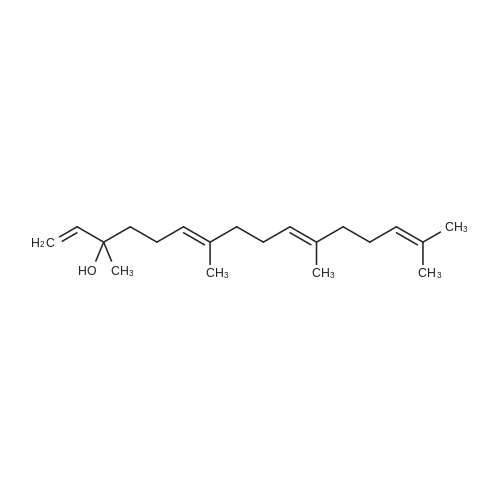
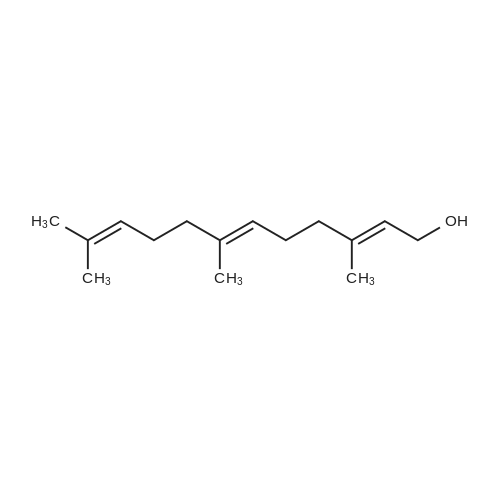
L-(+)-Diethyltartrate (428 muL, 2.5 mmol, 12.5 mol %) and 4A molecular sieves(2g, 0.1g/mmol) were placed in a 50 mL round bottom flask under a stream of argon.CH2Cl2 (20 mL) was added at room temperature, followed by Ti(Oi-Pr)4 (600 muL, 2mmol, 10 mol %). The mixture was stirred vigorously at room temperature for 20 min.tert-Butylhydroperoxide (4.54 mL, ~25 mmol, 125 mol%, 5-6 M in decane) was addedand the mixture was stirred 5 min at room temperature. The mixture was cooled in aCH3CN / dry ice bath. The temperature was maintained below -40 C. <strong>[106-28-5]Farnesol</strong> (5.06mL, 20 mmol, 100 mol %) was added and stirred in the CH3CN / dry ice bath for 10 h.The mixture was placed in the freezer overnight. The next day citric acid monohydrate(420 mg, 2 mmol, 10 mol%) was dissolved in 1:1 acetone / diethylether (~5 mL) and thesolution was added to the reaction mixture. The mixture was stirred vigorously for 20min at room temperature. Celite was added to the mixture and stirred vigorously for 1min. The slurry was filtered through a thick pad of celite and the celite was washed withEt2O. The clear filtrate was washed with saturated Na2S2O3 and then dried with MgSO4.Column chromatography isolated 4.53 g of S1 (95% yield). The enantiomeric excess wasdetermined by HPLC of the benzoate to be 87%. 1H NMR (400 MHz, CDCl3):delta 5.10 (m, 2H), 3.84 (ddd, J = 4.3, 7.5, 12.0 Hz, 1H), 3.70 (ddd, J = 4.9, 6.7, 11.8 Hz,1H), 2.99 (dd, J = 4.3, 6.7 Hz, 1H), 2.16-1.94 (m, 6H), 1.71 (m, 1H), 1.69 (s, 3H), 1.614(s, 3H), 1.608 (s, 3H), 1.48 (m, 1H), 1.32 (s, 3H).13C NMR (125 MHz, CDCl3):delta 136.0, 131.6, 124.4, 123.3, 63.2, 61.6, 61.4, 39.8, 38.7, 26.8, 25.9, 23.8, 17.9, 17.0,16.2.IR (NaCl, thin film): 3422, 2919, 1456, 1384, 1033 cm-1.HR-MS (ESI) m/z calcd for C15H26O2 [M+Na]+: 261.1825, found 261.1830.[alpha]20D = -4.2 (c = 1.9, CHCl3). Chiral HPLC analysis: Analysis was performed on the corresponding benzoate (BzCl,Et3N, DMAP, CH2Cl2): (Chiralcel AD-H, hexanes:2-propanol, 99:1, 1.0 mL/min):tR(2S,3S) = 7.3 min; tR(2R,3R) = 8.1 min. The enantiomeric excess was determined to be87%. |
| 54% |
With titanium(IV) isopropylate; tert.-butylhydroperoxide; diethyl (2R,3R)-tartrate; In dichloromethane; at -50℃; for 2h;Inert atmosphere; |
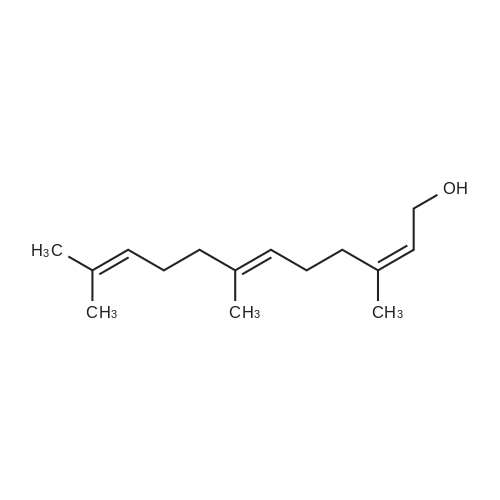
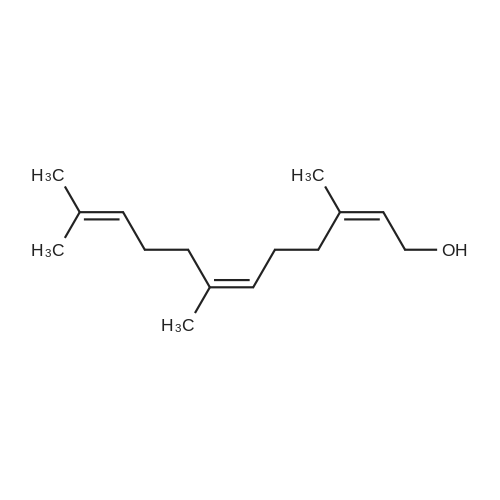
In a literature-known procedure [14], (+)-DET (1.13 g, 5.5 mmol, 1.1 equiv) and Ti(OiPr)4(1.54 g, 5.4 mmol, 1.08 equiv) were dissolved in CH2Cl2 (35 mL) and the mixture was cooledto - 50 C. A solution of farnesol (S1, 1.10 g, 5.0 mmol, 1.0 equiv) in CH2Cl2 (10 mL) wasadded dropwise, before tert-butyl hydroperoxide (5.5 M in decane, 2.0 mL, 11.0 mmol, 2.2equiv) was added. The reaction mixture was stirred for 2 h at -50 C and hydrolysed byaddition of 10% tartaric acid solution (10 mL). The reaction mixture was stirred for 2 h withoutcooling. The organic phase was washed with H2O (50 mL) and the aqueous phase wasextracted with CH2Cl2 (3 × 20 mL). The combined organic layers were dried with MgSO4 andthe solvent was removed under reduced pressure. The crude product was dissolved in Et2O (30 mL) and cooled to 0C, followed by addition of 1 M NaOH solution (15 mL). The reactionmixture was stirred for 40 min, the phases were separated and the aqueous phase wasextracted with Et2O (3 × 10 mL). The combined organic phases were washed with saturatedNH4Cl solution and brine followed by drying with MgSO4. The solvent was removed underreduced pressure. Column chromatography on silica gel [cyclohexane:EtOAc (8:1->5:1)]resulted in the (2S,3S) epoxy alcohol S2 (0.65 g, 2.7 mmol, 54%, >98% ee) as colourless oil.The same procedure was used to convert farnesol (10.0 mmol) to (2R,3R)-S2 (1.45 g, 6.1mmol, 61%, 86% ee) using (-)-DET. Mosher ester analyses [15] for the determination of theenantiomeric excesses were performed by dissolving 0.2 L of each epoxy alcohol in CDCl3(100 L). Then, pyridine (1 L) and (S)-MTPA-Cl (1 L) were added. The reaction mixturewas stirred for 30 min, diluted with CDCl3 (400 L) and analysed by 1H-NMR (Figure S5) |

 Chemistry
Chemistry
 Pharmaceutical Intermediates
Pharmaceutical Intermediates
 Inhibitors/Agonists
Inhibitors/Agonists
 Material Science
Material Science














 For Research Only
For Research Only
 110K+ Compounds
110K+ Compounds
 Competitive Price
Competitive Price
 1-2 Day Shipping
1-2 Day Shipping