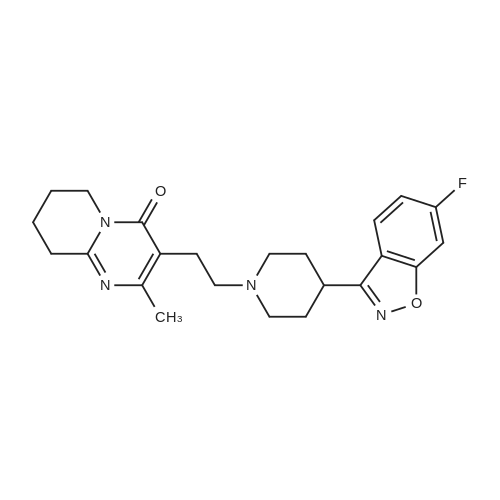
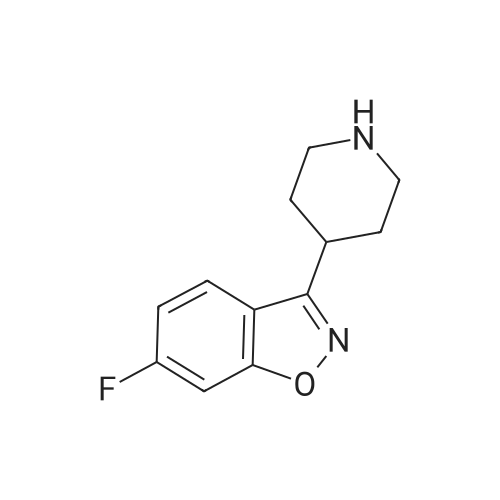
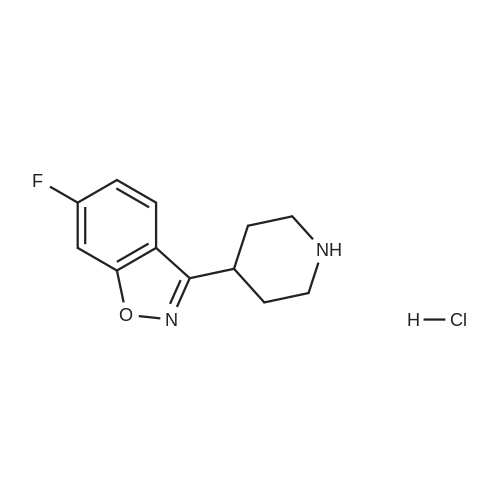
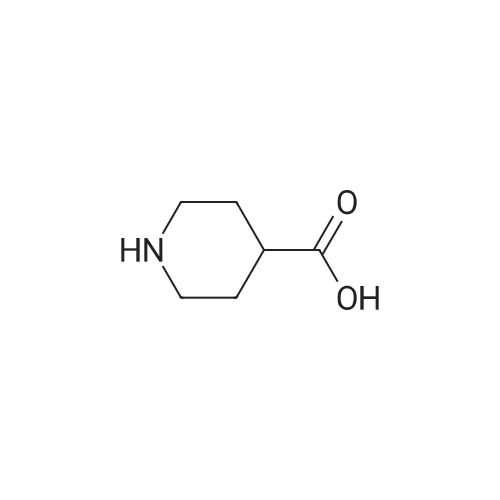
| 81% |
With sodium carbonate;potassium iodide; In acetonitrile; for 32h;Heating / reflux; |
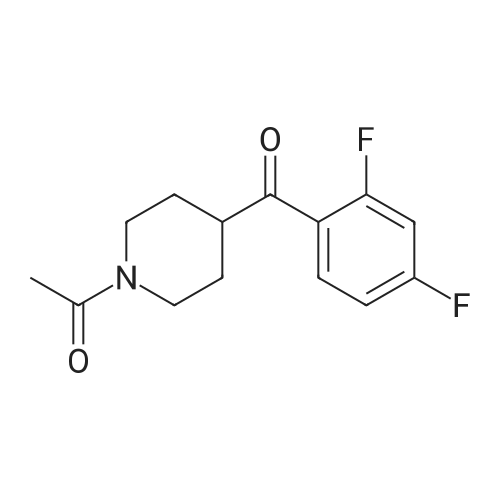
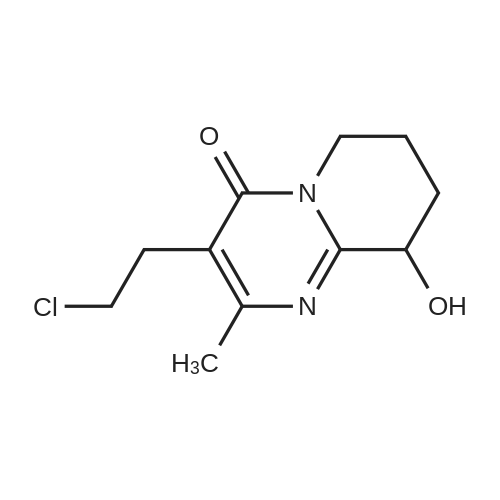
A mixture of 4.75 g of 3- (2-chloroethyl)-6, 7,8, 9-tetrahydro-2-methyl-4H-pyrido [[1,] [2-A] PYRIMIDIN-4-ONE] hydrochloride, 5.1 g of [4- (2,] 4-difluorobenzoyl) piperidine oxime hydrochloride, 0.46 g of potassium iodide, 5 g of anhydrous sodium carbonate and 30 ml of acetonitrile was stirred and refluxed for 32 hours. The reaction mixture was cooled and water (120 ml) was added under stirring. Separated solid stirred at [5 FOR 1] hour, filtered, washed with water and crystallized from ethyl acetate yielding 6 g (81%) of [3- [2- [4- (6-FLUORO-] [1,] [2-BENZISOXAZOLE-3-YL) PIPERIDINO] ETHYL]-6,] 7,8, [9-TETRAHYDRO-2-METHYL-4H-PYRIDO [ 1,] 2-a] [PYRIMIDIN-4-ONE.] |
| 79 - 81% |
With potassium carbonate;potassium iodide; In acetonitrile; for 30h;Heating / reflux; |
A mixture of 4.75 g of [3-(2-CHLOROETHYL)-6,] 7,8, 9-tetrahydro-2-methyl-4H-pyrido [1, 2-a] pyrimidin-4-one hydrochloride, 5. 1 g of [4- (2, 4-DIFLUOROBENZOYL)] piperidine oxime hydrochloride, 0.46 g of potassium iodide, 6.5 g of anhydrous powdered potassium carbonate and 30 ml of acetonitrile was stirred and refluxed for 30 hours. Reaction monitoring by HPLC analysis indicated in situ formation of risperidone. After the completion of reaction, the reaction mixture was cooled and water (120 ml) was added under stirring. Separated solid stirred at [5C] for [1] hour, filtered, washed with water and crystallized from ethyl acetate yielding 6 g [(81%) OF 3- [2- [4- (6-FLUORO-L,] 2-benzisoxazole-3-yl) piperidino] [ETHYL-6,] 7,8, 9- [TETRAHYDRO-2-METHYL-4H-PYRIDO [1,] 2-a] pyrimidin-4-one. Example-5 A mixture of 4.75 g of [3- (2-CHLOROETHYL)-6,] 7,8, 9-tetrahydro-2-methyl-4H-pyrido [[1,] 2-a] [PYRIMIDIN-4-ONE,] 6.0 g [OF 4- (2,] 4-difluorobenzoyl) piperidine oxime hydrochloride, [0.] 46 g of potassium iodide, 6.1 g of anhydrous powdered potassium carbonate and 40 ml of acetonitrile was stirred and refluxed for 30 hours. The reaction mixture was cooled and water (120 [ML)] was added under stirring. Separated solid stirred at [5C] for 1 hour, filtered, washed with water. Crude product was purified by crystallization in ethyl acetate to afford 6. [8] g (79%) of [3- [2- [4- (6-FLUORO-1, 2-BENZISOXAZOLE-3-YL) PIPERIDINO] ETHYL]-6,] 7,8, 9-tetrahydro-2- methyl-4H-pyrido [1, 2-a] [PYRIMIDIN-4-ONE.] |
| 77% |
With potassium carbonate;potassium iodide; In DMF (N,N-dimethyl-formamide); at 95 - 100℃; for 18h; |
A mixture of 4.75 g of [3.] [(2-CHLOROETHYL)-6,] 7,8, 9-tetrahydro-2-methyl-4H-pyrido [[1,] 2-a] [PYRIMIDIN-4-ONE] hydrochloride, [5.] [1] g of 4- (2, 4-difluorobenzoyl) piperidine oxime hydrochloride, 0.46 g of potassium iodide, 6.5 g of anhydrous powdered potassium carbonate and 30 ml [OF N, N-DIMETHYLFORMAMIDE] was stirred at [95-100C] for 18 hours. The reaction mixture was cooled and water (120 [ML)] was added under stirring. Separated solid stirred at [5C] for 1 hour, filtered, washed with. water and crystallized from ethyl acetate yielding 5.7 g [(77%)] of [3- [2- [4- (6-FLUORO-1,] 2-benzisoxazole-3-yl) piperidino] ethyl]-6, 7,8, 9-tetrahydro-2- [METHYL-4H-PYRIDO [1, 2-A] PYRIMIDIN-4-ONE.] |

 Chemistry
Chemistry
 Pharmaceutical Intermediates
Pharmaceutical Intermediates
 Inhibitors/Agonists
Inhibitors/Agonists
 Material Science
Material Science













 HazMat Fee +
HazMat Fee +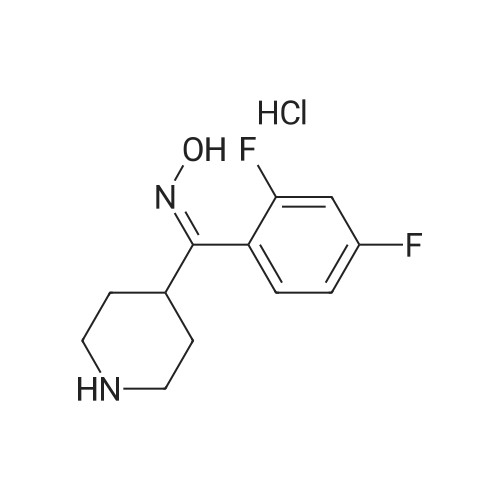

 For Research Only
For Research Only
 110K+ Compounds
110K+ Compounds
 Competitive Price
Competitive Price
 1-2 Day Shipping
1-2 Day Shipping