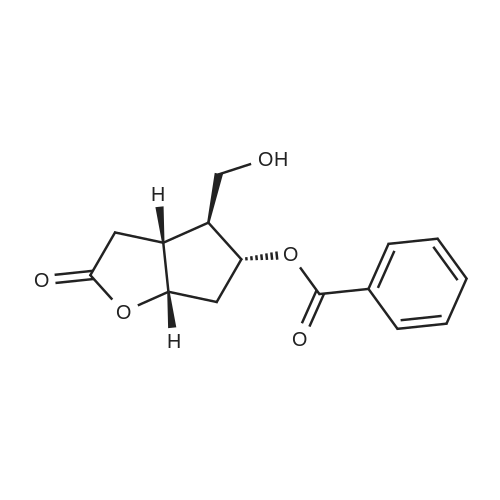
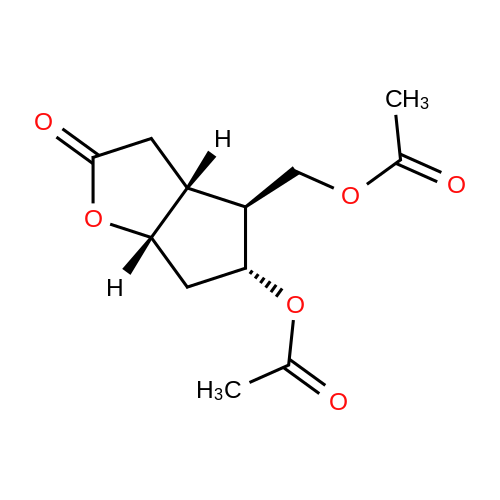
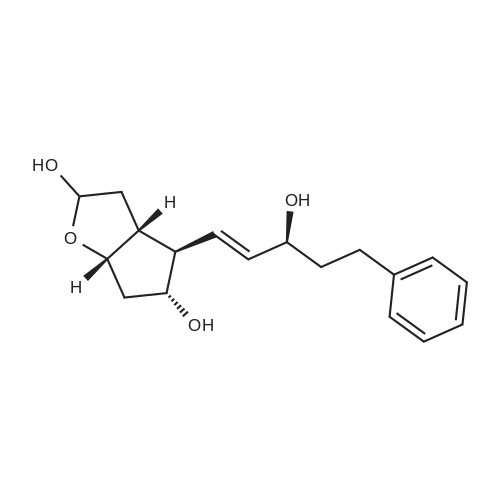
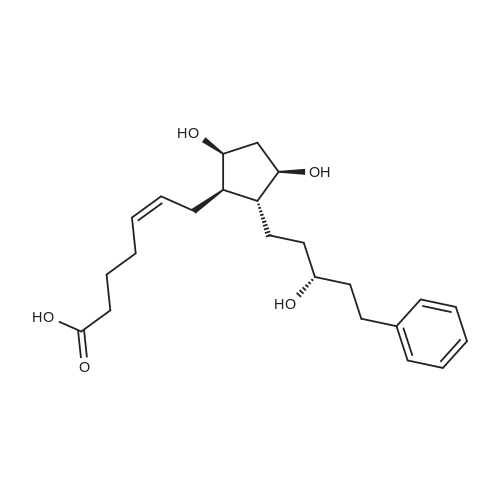
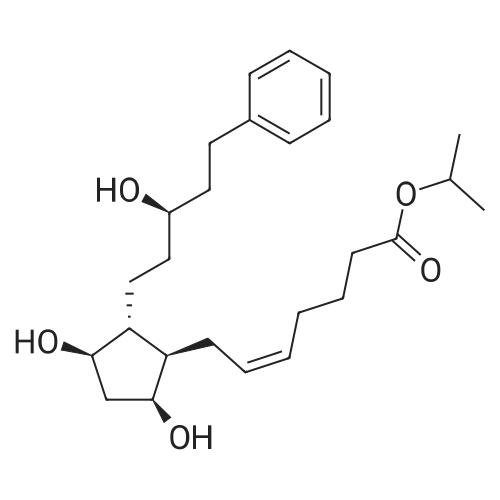
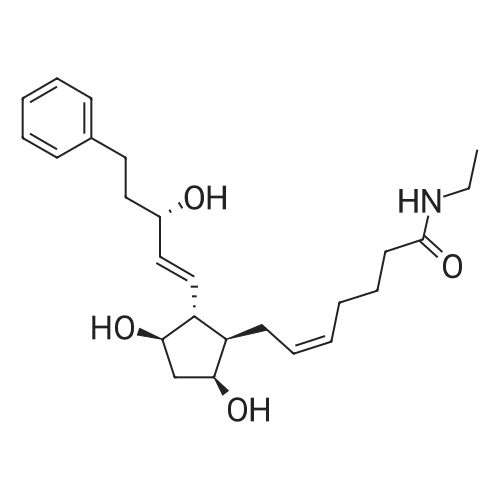
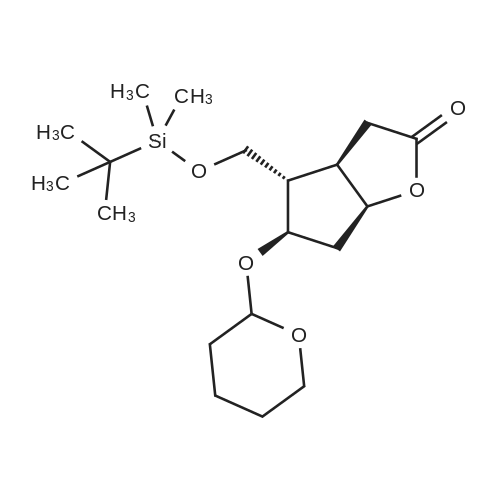
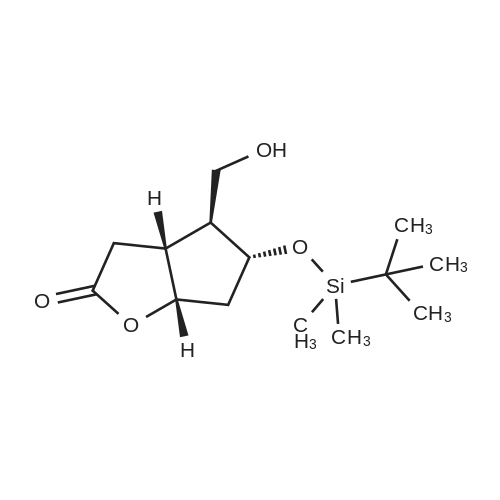
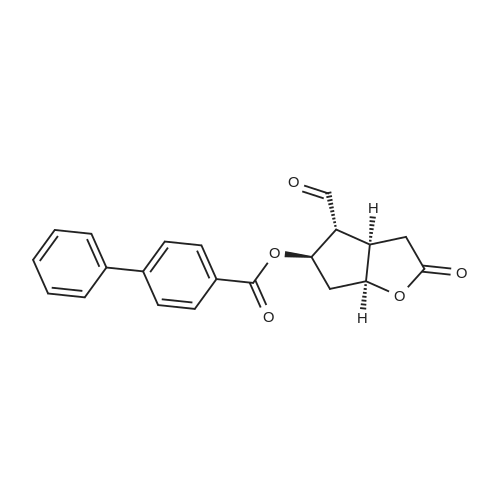
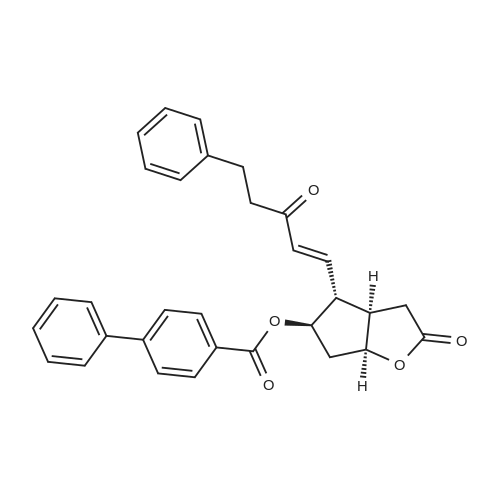
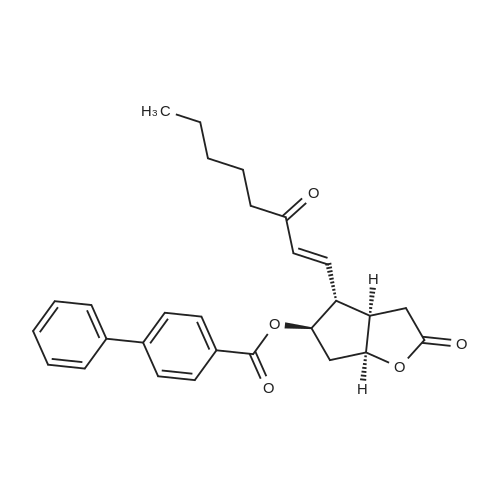
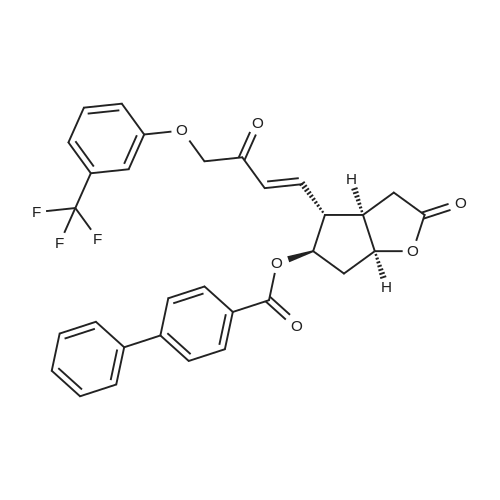
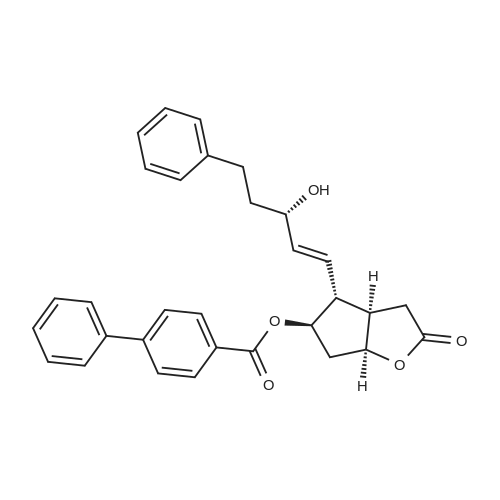
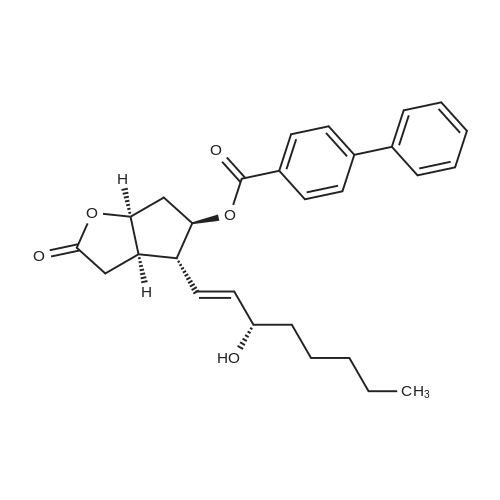
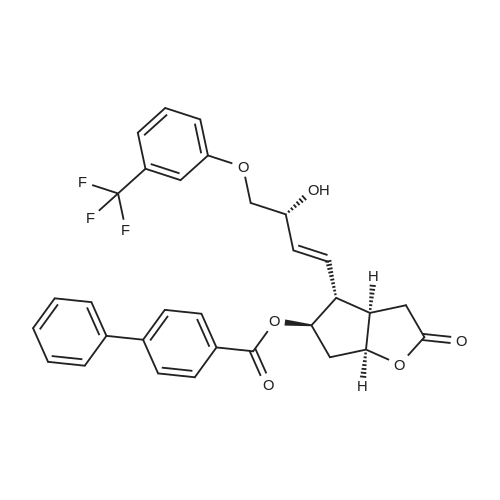
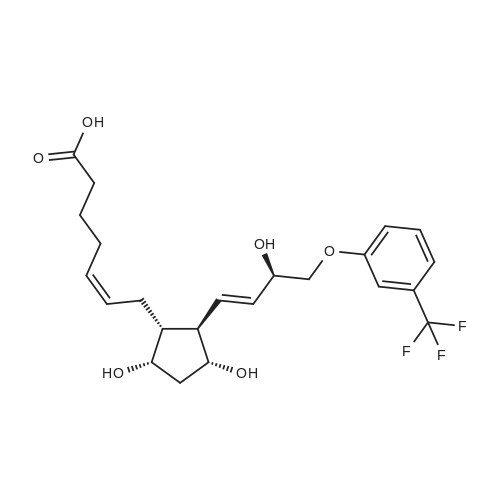
| 75% |
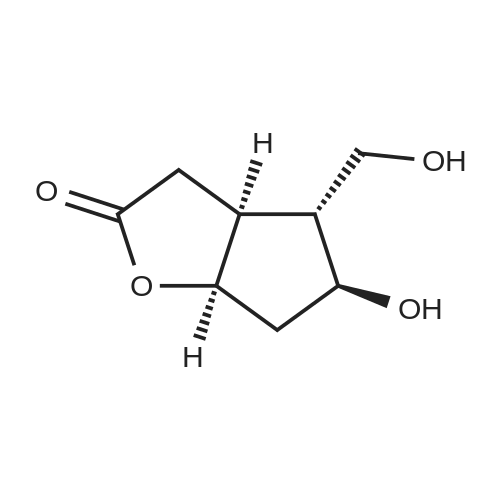
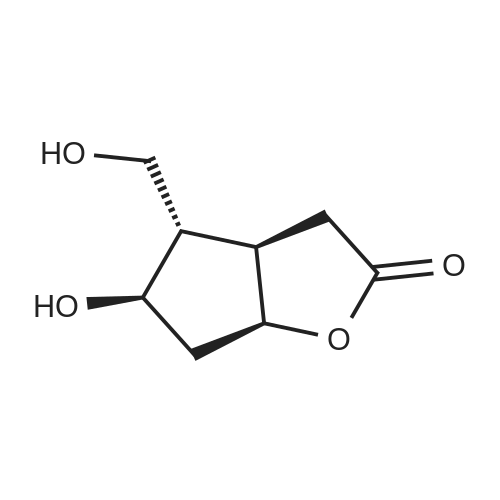
Stage #1: (3aR,4S,5R,6aS)-5-hydroxy-4-(hydroxymethyl)hexahydro-2H-cyclopenta[b]furan-2-one With 1H-imidazole In N,N-dimethyl-formamide at 20℃; for 0.333333h; Inert atmosphere;
Stage #2: tert-butyldimethylsilyl chloride In N,N-dimethyl-formamide for 8h; Inert atmosphere; |
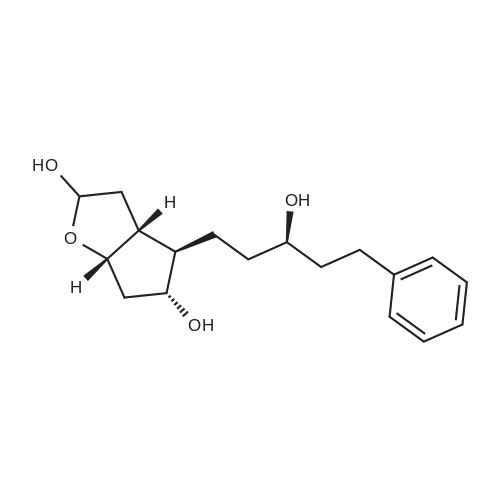
(3aR,4S,5R,6aS)-4- ((tert-butyldimethylsilyloxy)methyl)-5- hydroxyhexahydro-2H-cyclopenta[b] furan-2-one (4):3 To a solution of Corey lactone diol 3 (0.9 g, 5.23 mmol) in anhydrous DMF (1.8 mL) was added imidazole (0.88 g, 13.06 mmol). The solution was stirred for 20 min at room temperature under nitrogen atmosphere. Then tert-butyldimethylsilyl chloride (0.86 g, 5.74 mmol) was added in solution. The reaction mixture was stirred for 8 h, and then brine (~40 mL) was added to the reaction mixture and washed with EtOAc (3×10 mL). The organic layer was dried (Na2SO4) and the solvent was evaporated under reduced pressure. The crude product was purified by column chromatography eluting with ethyl acetate/hexane 1/1 to give the white solid 4 (1.11 g, 75%). Rf 0.4 (EtOAc/Hex 1/1); m.p. 61 ºC; [α]D -14.9 (c. 1.05, CHCl3) IR (KBr) cm-1: 3370.1, 2940.81, 2885.8, 1739.37. 1H NMR (500 MHz, CDCl3) δ= 4.9 (dt, J = 6.7 Hz, 1H), 4.15 (q, J = 10.0, Hz, 1H), 3.7 (d, J = 12.4 Hz, 1H), 3.6 (d, J = 12.4 Hz, 1H), 2.8 (m, 1H), 2.65-2.3 (m, 3H), 2.0 (m, 2H), 0.89 (s, 9H), 0.06 (s, 6H) ppm. 13C NMR (125 MHz, CDCl3) δ= -5.5 (2xCH3), 1.8 (C), 25.8 (3xCH3), 35.3 (CH2), 39.6 (CH), 40.8 (CH2), 55.3 (CH), 63.9 (CH2), 75.5 (CH), 83.8 (CH), 177 (C=O) ppm. MS [EI+] m/z (%): 287 (M++1), 229 (15), 183 (12), 137 (47), 75 (100), 41 (7). |
|
With 1H-imidazole In N,N-dimethyl-formamide at -15 - 0℃; Inert atmosphere; |
1
EXAMPLE 1 (3aR,4S,5R,6aS)-4-[(tert-butyldimethylsilyloxy)methyl]-5-(tert-butyldiphenyl-silyloxy) hexahydro-2H-cyclopenta[b]furan-2-one [Show Image] In a round bottom flask (3aR,4S,5R,6aS)-5-hydroxy-4-(hydroxymethyl)hexahydro-2H-cyclopenta[b]furan-2-one (100.0 g, 0.581 mol) was dissolved in anhydrous N,N-dimethylformamide (500 mL) under nitrogen atmosphere. The flask was cooled to 15°C and imidazole (42.31 g, 0.621 mol) was added followed by tert-butyldimethylsilyl chloride (91.91 g, 0.619 mol). The mixture was stirred at -13°C for one hour, then the temperature was allowed to reach 0°C and stirred overnight. After checking the selective protection of the primary hydroxyl group by TLC analysis, imidazole (63.26 g, 0.929 mol) and tert-butyl(chloro)diphenylsilane (175.2 mL, 185.19 g, 0.674 mol) were added at 0°C and the mixture was stirred at the same temperature for one hour and subsequently at room temperature. The reaction was checked by TLC and quenched by addition of ethyl acetate (1.00 L), water (1.00 L) and 5% sodium bisulfate solution (300 mL). The phases were separated and the aqueous one was extracted with ethyl acetate (330 mL). The combined organic layers were washed with water (3x 330 mL), brine (460 mL, 160 mL) and dried over magnesium sulfate. After filtration the organic solvent was removed at 40°C under reduced pressure. The crude product was obtained as a yellow oil (357.88 g) and was used in the subsequent step without further purification. A sample (5.50 g, corresponding to 0.009 mol of starting diol) was purified by column chromatography on silica gel (eluent: n-hexane: ethyl acetate from 95:5 to 80:20) affording the product (4.50 g, 0.0086 mol, 95%) as a colorless oil which was used for the characterization. 1H-NMR {400MHz, CDCl3, δ (ppm)}: 7.25-7.65 (m, 10 H, Ph), 4.84 (m, 1H, CH-O-C=O), 4.09 (m, 1H, CH-O-Si), 3.40 (dd, J=5.2, 10.0Hz, 1H, CH2-O-Si), 3.27 (dd, J=6.0, 10.0Hz, 1H, CH2-O-Si), 2.80 (dd, J=11.2, 18.8 Hz, 1H, CH2-C=O), 2.64 (m, 2H, -O-CH-CH2-CH-O+ CH2-C=O), 1.90-2.10 (m, 3H, -O-C H-CH2-CH-O+CH+CH), 1.04 (s, 9H, (CH3)3C), 0.79 (s, 9H, (CH3)3C), 0.08 (s, 3H, (CH3)Si), 0.07 (s, 3H, (CH3)Si). 13C-NMR {400MHz, CDCl3, δ (ppm)}: 177.2 (C), 135.8 (2x arom.CH), 135.7 (2x arom.CH), 133.6 (C), 133.5 (C), 129.8 (arom.CH), 129.7 (arom.CH), 127.6 (2x arom.CH), 127.5 (2x arom.CH), 84.2 (CH), 76.2 (CH), 63.2 (CH2), 57.1 (CH), 41.0 (CH2), 39.8 (CH), 35.8 (CH2), 26.8 (3x CH3), 25.7 (3x CH3), 18.9 (C), 18.0 (C), -5.7 (2x CH3). HPLC-MS (ESI): [M-(t-Bu(CH3)3Si) +H2O]+ = 428; [M-(t-Bu(CH3)3Si)+Na]+ = 433. |
|
With dmap; triethylamine In dichloromethane at -5 - 25℃; for 14h; |
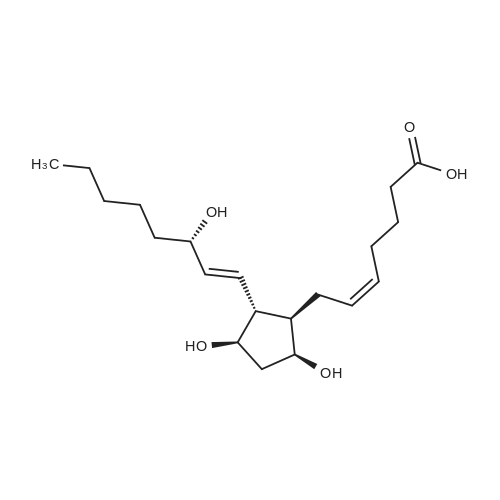
(Preparation of starting materials): Compound (IV) used in the present invention: 7-[(1R,2R,3R,5S)-2-(4,4-difluoro-3-hydroxyoctyl)-5-hydroxy-3-(2-tetrahydrogen) Pyryloxy)cyclopentyl]heptanoic acid is prepared by the following method:
At -5°C,Slowly add 125 mL of dichloromethane solution containing 43.7 g of TBSCl to 50 g of compound 1,375 mL of dichloromethane, 29.4 g of a mixture of triethylamine and 3.57 g of DMAP.After the addition was complete, the mixture was stirred for 2.0 hours.Warm to 25°C and stir for 12 hours.The reaction was complete, and 175 ml of water was added to the reaction solution.After the addition is complete, stir for 10-20 minutes to separate the organic phase.It was washed with saturated sodium chloride solution, dried over anhydrous sodium sulfate, filtered and concentrated under reduced pressure to give compound 2 as 88.89 g of an oily product. |

 Chemistry
Chemistry
 Pharmaceutical Intermediates
Pharmaceutical Intermediates
 Inhibitors/Agonists
Inhibitors/Agonists
 Material Science
Material Science













 For Research Only
For Research Only
 110K+ Compounds
110K+ Compounds
 Competitive Price
Competitive Price
 1-2 Day Shipping
1-2 Day Shipping