| 94% |
With acetic acid In ethanol at 75 - 80℃; |
Example-2
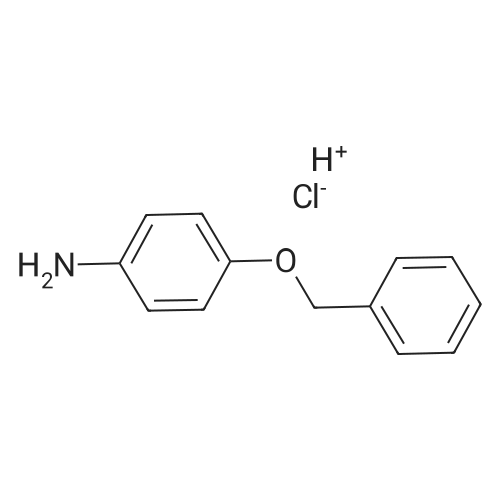
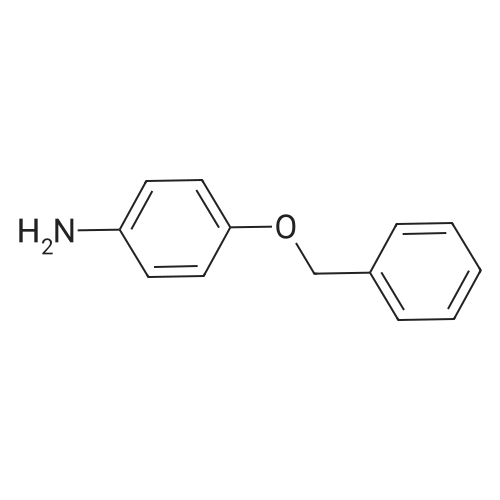
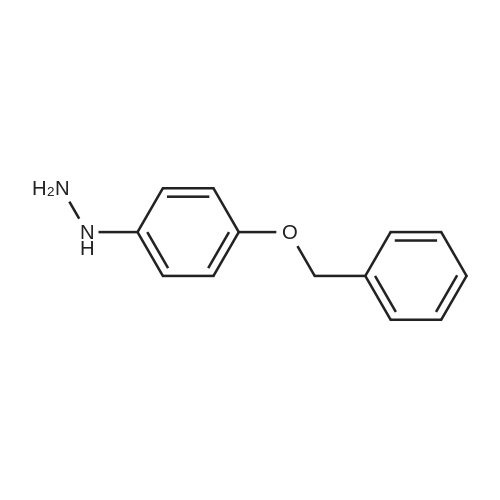
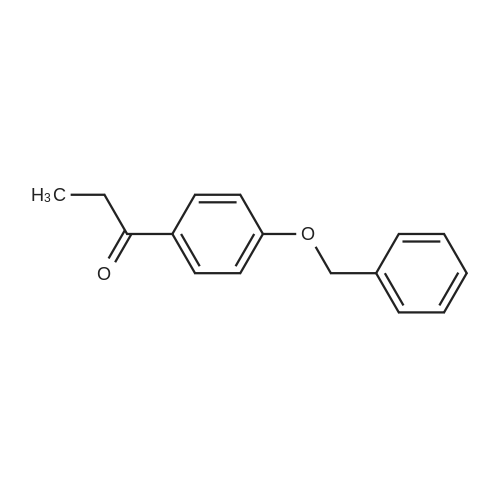
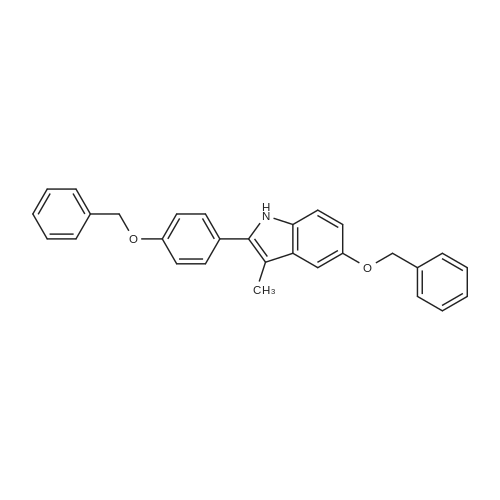
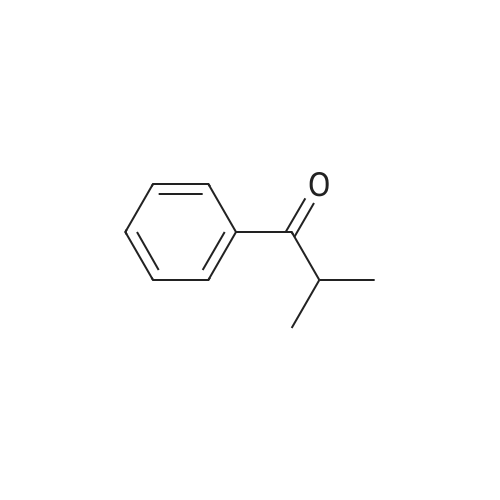
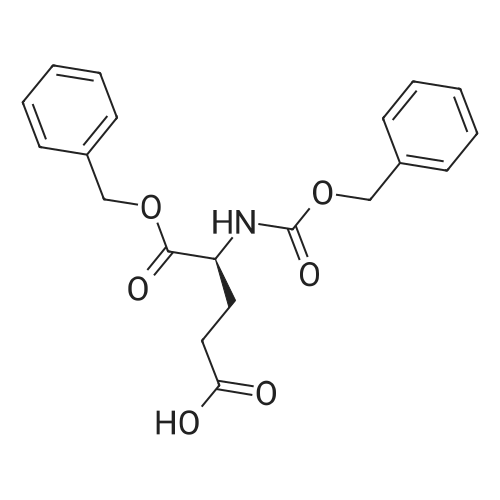
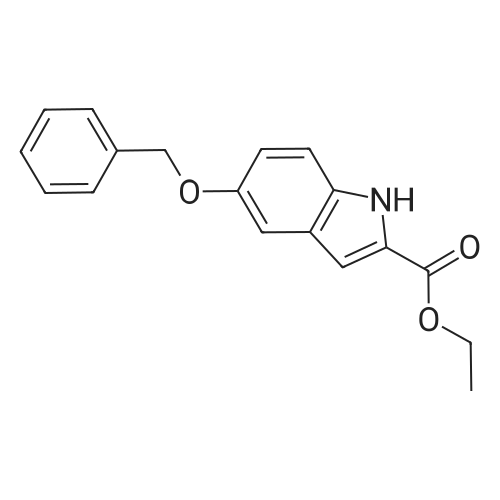
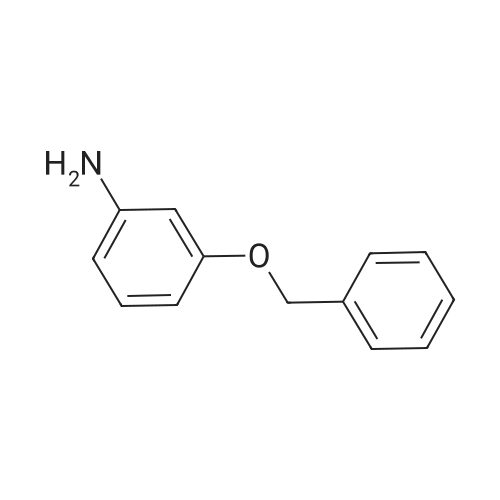
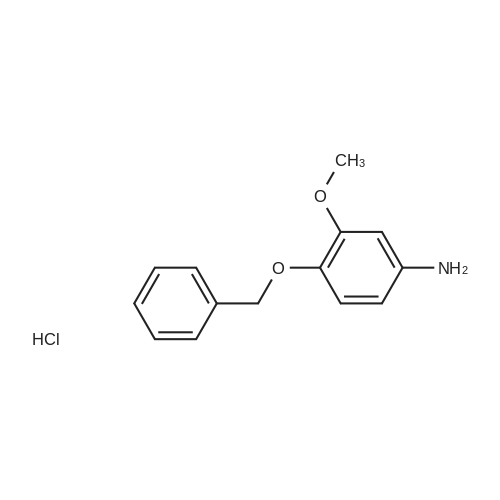
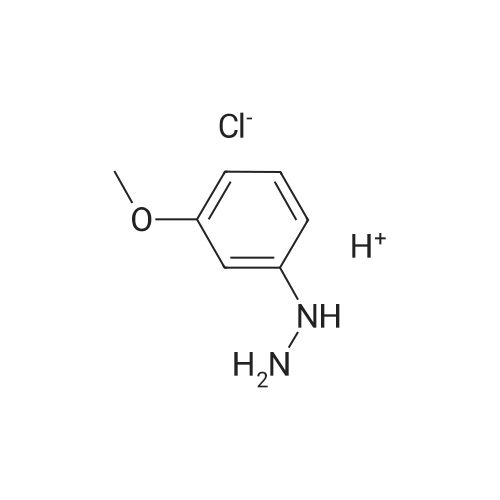
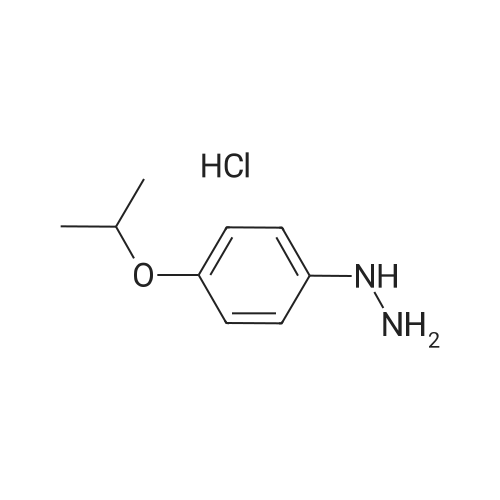
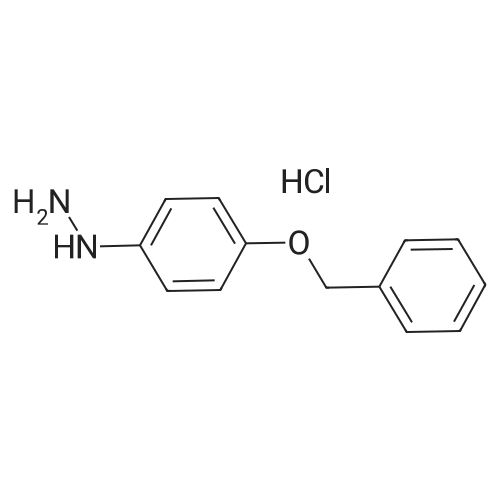
4-Benzyloxy phenyl hydrazine hydrochloride (10 g, 40 mmol), 4-benzyloxy propiophenone (9.6 g, 40 mmol) and acetic acid (0.1 ml, 1.7 mmol) were suspended in 140 ml of ethanol and the mixture was refluxed for 12 hrs at a temperature of 75-80° C.
During said period the product precipitates.
After cooling the mixture at 10-15° C. the crystallized product was isolated by filtration and washed with chilled ethanol (30 ml) and water (50 ml).
5-Benzyloxy-2-(4-benzyloxy)-3-methyl-1H-indole melting at 152-153° C. was obtained in a yield of 15.7 g (94percent).
1H-NMR (DMSO) δ 10.65 (s, 1H); 7.55 (d, 2H); 7.50 (d, 4H); 7.30-7.45 (m, 6H); 7.21 (d, 1H); 7.10 (d, 1H); 7.10 (d, 1H); 6.91 (dd, 1H); 5.16 (s, 2H); 5.11 (s, 2H); 2.33 (s, 3H) |
| 94% |
at 75 - 80℃; for 12 h; |
Example-2
4-Benzyloxy phenyl hydrazine hydrochloride (10 g, 40 mmol), 4-benzyloxy propiophenone (9.6 g, 40 mmol) and acetic acid (0.1 ml, 1.7 mmol) were suspended in 140 ml of ethanol and the mixture was refluxed for 12 hrs at the temperature of 75 to 80°C.
During said period the product precipitates.
After cooling the mixture at 10 to 15°C the crystallized product was isolated by filtration and washed with chilled ethanol (30 ml) and water (50 ml).
5-Benzyloxy-2-(4-benzyloxy)-3-methyl-1H-indole melting at 152 to 153°C was obtained in a yield of 15.7 g (94percent).
1H-NMR (DMSO) δ 10.65 (s, 1H); 7.55 (d, 2H); 7.50(d, 4H); 7.30-7.45(m, 6H); 7.21(d, 1H); 7.10(d, 1H); 7.10(d, 1H); 6.91(dd, 1H); 5.16(s, 2H); 5.11(s, 2H); 2.33(s, 3H) |
| 84% |
With hydrogenchloride In ethanol for 2 h; Reflux |
1-(4-(Benzyloxy)phenyl)propan-1-one (2) (1.9 g, 8.0 mmol) and1-(4-(benzyloxy)phenyl)hydrazine hydrochloride (4) (2.0 g,8.0 mmol) were dissolved in EtOH (50 mL) and 5 drops of conc.HCl was added and the mixture was refluxed for 2 h. The reaction mixture was then allowed to cool and was stirred for 1 h. The mixturewas filtered to afford an pale pink solid. The solid was washedwith cold EtOH (10 mL) followed by n-hexane (20 mL). (2.8 g, 84percent).Mp: 149–152 C.50 IR: mmax (KBr) cm1: 3470 (N–H), 2880, 2820,1620 (CC). 1H NMR (CDCl3, 400 MHz) d 7.86 (br s, 1H, NH),7.48–7.58 (m, 6H, Ar-H), 7.33–7.47 (m, 6H, Ar-H), 7.25–7.31 (m,1H, Ar-H), 7.14–7.18 (m, 1H, Ar-H), 7.07–7.13 (m, 2H, Ar-H), 6.97(d, J = 7.53 Hz, 1H, Ar-H), 5.18 (s, 2H, CH2), 5.15 (s, 2H CH2), 2.43(s, 3H, CH3) 13C NMR (CDCl3, 100 MHz) d 157.7 (BnO–C), 157.3(BnO–C), 143.6 (Ar-C–CH2), 137.3 (Cq), 136.3 (Cq), 136.0 (Cq),129.9 (ArC), 128.5 (ArC), 128.2 (ArC), 128.2 (ArC), 127.7 (ArC),127.6 (Ar-C), 127.1 (ArC), 120.4 (ArC), 114.7 (ArC), 114.6 (ArC),114.3 (ArC), 112.2 (Cq), 110.8 (CC), 108.8 (ArC), 102.0 (ArC),70.0 (CH2), 69.6 (CH2), 9.3 (CH3). HRMS (EI): Found 442.1797 (M+Na)+, C29H25NNaO2 requires 442.1783. |

 Chemistry
Chemistry
 Pharmaceutical Intermediates
Pharmaceutical Intermediates
 Inhibitors/Agonists
Inhibitors/Agonists
 Material Science
Material Science














 For Research Only
For Research Only
 110K+ Compounds
110K+ Compounds
 Competitive Price
Competitive Price
 1-2 Day Shipping
1-2 Day Shipping