Alternatived Products of [ 104317-95-5 ]
Product Details of [ 104317-95-5 ]
| CAS No. : | 104317-95-5 |
MDL No. : | MFCD11520725 |
| Formula : |
C7H6ClIO
|
Boiling Point : |
- |
| Linear Structure Formula : | - |
InChI Key : | - |
| M.W : |
268.48
|
Pubchem ID : | - |
| Synonyms : |
|
Safety of [ 104317-95-5 ]
| Signal Word: | |
Class: | |
| Precautionary Statements: | |
UN#: | |
| Hazard Statements: | |
Packing Group: | |
Application In Synthesis of [ 104317-95-5 ]
* All experimental methods are cited from the reference, please refer to the original source for details. We do not guarantee the accuracy of the content in the reference.
- Upstream synthesis route of [ 104317-95-5 ]
- Downstream synthetic route of [ 104317-95-5 ]
- 1
-
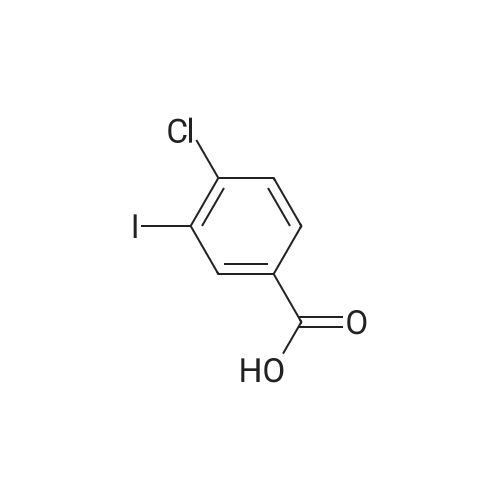 [ 42860-04-8 ]
[ 42860-04-8 ]

-
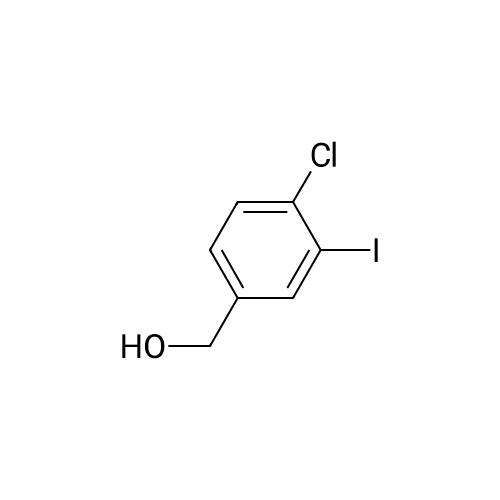 [ 104317-95-5 ]
[ 104317-95-5 ]
| Yield | Reaction Conditions | Operation in experiment |
|
With sodium hydroxide; In tetrahydrofuran; methanol; ethyl acetate; |
(i)4-chloro-3-iodobenzyl Alcohol Borane-THF complex (10 ml) was added dropwise over 20 minutes to a solution of <strong>[42860-04-8]4-chloro-3-iodobenzoic acid</strong> (1.4 g) in THF (25 ml). The reaction mixture was stirred for 2 hours and then cooled (ice bath) and methanol (20 ml) was added cautiously. The solvent was removed and the residue was dissolved in methanol (10 ml) and stirred with aq. 2M sodium hydroxide (10 ml) for 2 hours. Ethyl acetate (50 ml) was added and the mixture was washed with saturated aq. sodium bicarbonate solution (50 ml). The aqueous extracts were washed with ethyl acetate (2*50 ml) and the combined ethyl acetate extracts were washed with water (50 ml) and brine (50 ml) and died. Removal of the solvent gave 4-chloro-3-iodobenzyl alcohol (1.15 g). NMR (CD3SOCD3): d 4.45 (d, 2H), 5.3 (t, 1H), 7.3 (m, 1H), 7.5 (m, 1H), 7.8 (s, 1H). |
- 2
-
 [ 104317-95-5 ]
[ 104317-95-5 ]

-
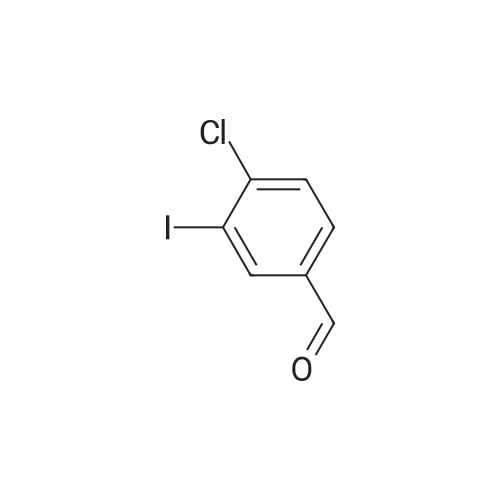 [ 276866-90-1 ]
[ 276866-90-1 ]
- 3
-
aqueous sodium bisulphite
[ No CAS ]

-
 [ 42860-04-8 ]
[ 42860-04-8 ]

-
 [ 104317-95-5 ]
[ 104317-95-5 ]
| Yield | Reaction Conditions | Operation in experiment |
|
With hydrogenchloride; sodium borohydrid; iodine; In tetrahydrofuran; |
a) Preparation of 2-chloro-1-iodo-5-hydroxymethylbenzene according to J. Org. Chem., 1991, 56, 5964-5965, from the corresponding commercial benzoic acid. 5.02 g of sodium borohydride are added portionwise and then 14.6 g of iodine, in solution in 50 ml of tetrahydrofuran, are added very slowly to 25 g of <strong>[42860-04-8]4-chloro-3-iodobenzoic acid</strong> in solution in 200 ml of tetrahydrofuran at 0° C. The reaction mixture is stirred for 2 hours at room temperature and then at 35° C. for 30 minutes. Hydrolysis is carried out at 10° C. with a 0.5N hydrochloric acid solution and extraction is carried out with ethyl acetate. The organic phase is separated by settling and then treated with an aqueous sodium bisulphite solution and then with water. The organic phase is dried over anhydrous sodium sulphate and the solvents are evaporated under reduced pressure. The expected compound is obtained by distillation; B.p.=109° C. under 3 Pa. |

 Chemistry
Chemistry
 Pharmaceutical Intermediates
Pharmaceutical Intermediates
 Inhibitors/Agonists
Inhibitors/Agonists
 Material Science
Material Science















 For Research Only
For Research Only
 110K+ Compounds
110K+ Compounds
 Competitive Price
Competitive Price
 1-2 Day Shipping
1-2 Day Shipping





