| 97% |
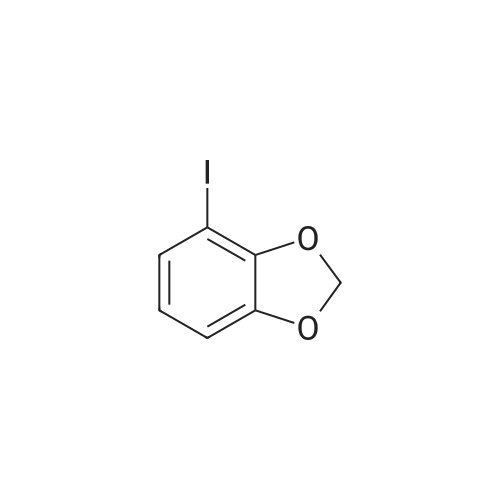
Stage #1: Sesamol With sodium hydride In tetrahydrofuran; N,N-dimethyl-formamide at 0 - 20℃;
Stage #2: chloromethyl methyl ether In tetrahydrofuran; N,N-dimethyl-formamide at 0 - 20℃; Further stages.; |
|
| 96% |
With sodium hydride In diethyl ether; N,N-dimethyl-formamide at 0 - 20℃; |
|
| 96% |
Stage #1: Sesamol With sodium hydride In tetrahydrofuran; N,N-dimethyl-formamide at 0℃; for 0.833333h; Inert atmosphere;
Stage #2: chloromethyl methyl ether In tetrahydrofuran; N,N-dimethyl-formamide at 20℃; for 1h; Inert atmosphere; |
|
| 91% |
With N-ethyl-N,N-diisopropylamine In dichloromethane |
|
| 91% |
Stage #1: Sesamol With sodium hydride In tetrahydrofuran at 0 - 20℃; for 0.5h; Inert atmosphere;
Stage #2: chloromethyl methyl ether In tetrahydrofuran at 0 - 20℃; for 16h; Inert atmosphere; |
5. General synthetic procedure for intermediate 19
A solution of sesamol (16, 10.0 g, 72.40 mmol) in dry THF (50 mL) was slowly added to a stirred solution of NaH (4.09 g, 102.25 mmol) in dry THF (50 mL) at 0 under N2 atmosphere. After the mixture was stirred for 0.5 h at room temperature. The reaction mixture was cooled again to 0 and MOMCl (5.77 mL, 76.02 mmol) was added dropwise to the above mixture. After completing addition, the reaction mixture was stirred for 16 h at room temperature. The resulting reaction mixture was quenched with ice-water (200 mL) and extracted with EtOAc (150 mL). The combined organic layer was washed with brine (200 mL), dried over anhydrous Na2SO4, filtered and concentrated under reduced pressure. The residue was purified by silica gel chromatography eluting with ethyl acetate/petroleum ether (1:100-1:50, v/v) to afford the intermediate (17, 11.95 g, 91% yield). 1H NMR (400 MHz, CDCl3) δ 6.70 (d, J = 8.5 Hz, 1H), 6.63 (d, J = 2.4 Hz, 1H), 6.49 (dd, J = 8.5, 2.4 Hz, 1H), 5.91 (s, 2H), 5.08 (s, 2H), 3.47 (s, 3H). 13C NMR (101 MHz, CDCl3) δ 152.62 (s), 148.20 (s), 142.62 (s), 108.52 (s), 108.10 (s), 101.28 (s), 99.81 (s), 95.56 (s), 55.95 (s). |
| 90% |
With sodium hydride In diethyl ether; N,N-dimethyl-formamide at 0℃; |
|
| 90% |
Stage #1: Sesamol With sodium hydride In tetrahydrofuran; N,N-dimethyl-formamide at 0 - 20℃; for 0.5h; Inert atmosphere;
Stage #2: chloromethyl methyl ether In tetrahydrofuran; N,N-dimethyl-formamide at 0 - 20℃; for 0.5h; Inert atmosphere; |
|
| 78% |
With N-ethyl-N,N-diisopropylamine In dichloromethane at 0 - 20℃; for 21h; Inert atmosphere; |
3 5-Methoxymethoxy-1,3-benzodioxole (S4)
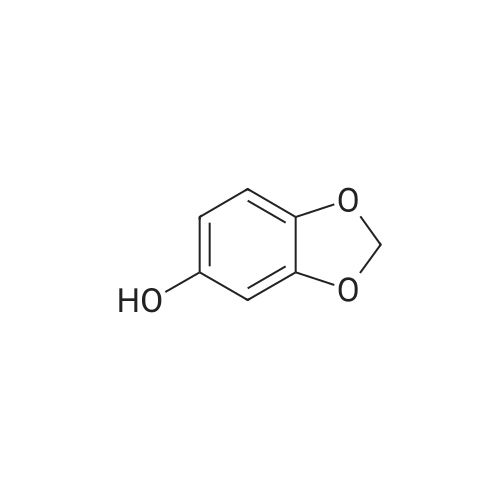
General procedure: Under an argon atmosphere,chloromethyl methyl ether (1.98 mL, 26.0 mmol) was added dropwise to a solution of 2-bromophenol (3.00 g, 17.3 mmol) and N,N-diisopropylethylamine (6.04 mL, 34.7mmol) in dichloromethane (60 mL) at 0 °C. After stirring for 1 h, the reaction mixture was allowed to warm to room temperature and stirred for an additional 20 h. The mixture was quenched with saturated aqueous ammonia and the products were extract with dichloromethane.The extract was washed with 10% aqueous NaOH, water, and brine, dried over Na2SO4, and evaporated. The residue was purified by bulb-to-bulb distillation (95-110 °C, 5 mmHg) to give S1 as colorless oil (3.58 g, 95%). |
|
With sodium hydride 1.) DMF, 80 deg C, 1h, 2.) from 0 deg C to rt overnight; Yield given. Multistep reaction; |
|
|
With N-ethyl-N,N-diisopropylamine In dichloromethane at 0 - 20℃; |
|
| 4.7 g |
With N-ethyl-N,N-diisopropylamine In dichloromethane at 0 - 20℃; |
|
| 17.17 g |
With N-ethyl-N,N-diisopropylamine In tert-butyl methyl ether at 0 - 25℃; |
|
|
Inert atmosphere; |
|
|
Stage #1: Sesamol With sodium hydride In tetrahydrofuran; mineral oil at 0℃; for 1h;
Stage #2: chloromethyl methyl ether In tetrahydrofuran; mineral oil at 0 - 20℃; for 1h; |
6.1 Step 1: Example 6b
To a solution of Example 6a (6.1 g, 44.2 mmol) in THF (60 mL) was added NaH partially (60%, 2.6 g, 66.3 mmol) at 0 °C, the mixture was stirred at 0 °C for lh, and then MOM-C1 (4.2 g, 53.1 mmol) was added slowly at 0 °C. After adding over, the reaction was then turned to room temperature and stirred for another 1 hr. The reaction was quenched by adding 90 mL water and extracted with EtOAc (100 mL*2). The combined organic phase was washed with brine, dried over Na2S04, and concentrated under residued pressure to give 10.1 g of yellow oil, which was further purified by silica gel chromatography to give the desired product (Example 6b, 4.2 g , crude yield 52%) as colorless oil. MS [M+13]+= 195.1. |

 Chemistry
Chemistry
 Pharmaceutical Intermediates
Pharmaceutical Intermediates
 Inhibitors/Agonists
Inhibitors/Agonists
 Material Science
Material Science














 For Research Only
For Research Only
 110K+ Compounds
110K+ Compounds
 Competitive Price
Competitive Price
 1-2 Day Shipping
1-2 Day Shipping