Alternatived Products of [ 1251041-55-0 ]
Product Details of [ 1251041-55-0 ]
| CAS No. : | 1251041-55-0 |
MDL No. : | MFCD30292390 |
| Formula : |
C11H6ClNO
|
Boiling Point : |
- |
| Linear Structure Formula : | - |
InChI Key : | - |
| M.W : |
203.62
|
Pubchem ID : | - |
| Synonyms : |
|
Safety of [ 1251041-55-0 ]
| Signal Word: | |
Class: | N/A |
| Precautionary Statements: | |
UN#: | N/A |
| Hazard Statements: | |
Packing Group: | N/A |
Application In Synthesis of [ 1251041-55-0 ]
* All experimental methods are cited from the reference, please refer to the original source for details. We do not guarantee the accuracy of the content in the reference.
- Upstream synthesis route of [ 1251041-55-0 ]
- Downstream synthetic route of [ 1251041-55-0 ]
- 1
-
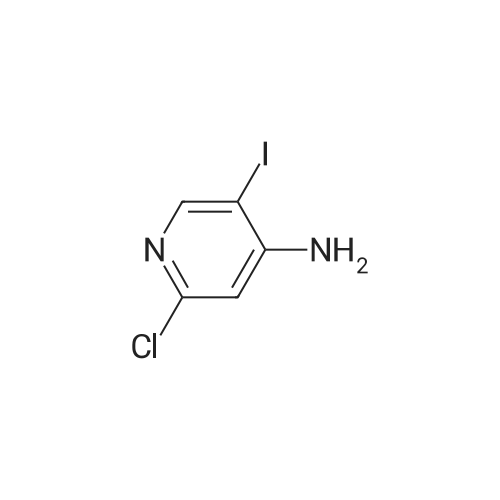 [ 800402-12-4 ]
[ 800402-12-4 ]

-
potassium phosphate
[ No CAS ]

-
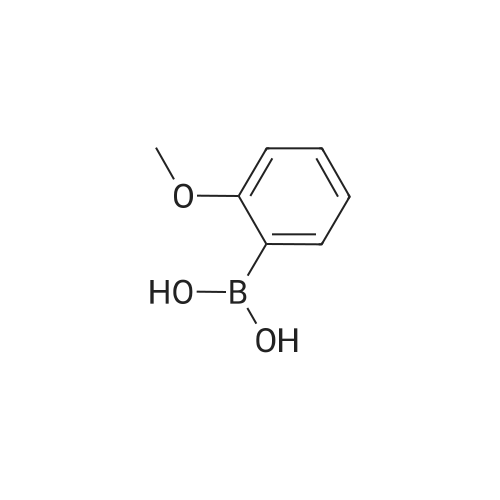 [ 5720-06-9 ]
[ 5720-06-9 ]

-
 [ 603-35-0 ]
[ 603-35-0 ]

-
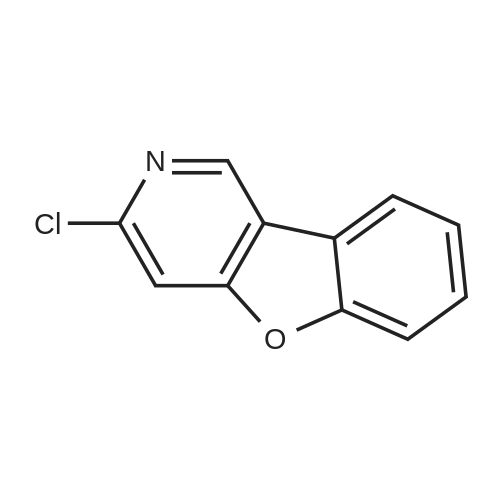 [ 1251041-55-0 ]
[ 1251041-55-0 ]
| Yield | Reaction Conditions | Operation in experiment |
|
palladium diacetate; In water; acetonitrile; |
Potassiumphosphate (18.28 g, 79 mmol), triphenyl phosphine (1.04 g, 3.97 mmol), <strong>[800402-12-4]2-chloro-5-iodo-4-aminopyridine</strong> (10.1 g, 39.mmol), 2-methoxybenzeneboronic acid (8.44 g, 55.57 mmol) and palladium acetate (0.45 g, 1.98 mmol) were sequentially added to degassed acetonitrile (300 mL) and water (100 mL) under nitrogen. The reaction mixture was heated at 75 C. for overnight, then cooled to room temperature. The organic layer was separated and aqueous layer and was extracted with ethyl acetate. The combined organic layers were dried over sodium sulfate, concentrated and performed column chromatography using hexanes and ethyl acetate as eluent. 7.5 g of title compound was isolated. Synthesis of 3-chlorobenzofuro[3,2-c]pyridine: |
- 2
-
 [ 657408-07-6 ]
[ 657408-07-6 ]

-
potassium phosphate
[ No CAS ]

-
Pd2(bda)3
[ No CAS ]

-
 [ 1251041-55-0 ]
[ 1251041-55-0 ]

-
 [ 98-80-6 ]
[ 98-80-6 ]

-
 [ 800402-12-4 ]
[ 800402-12-4 ]
| Yield | Reaction Conditions | Operation in experiment |
|
With nitrogen; In water; toluene; |
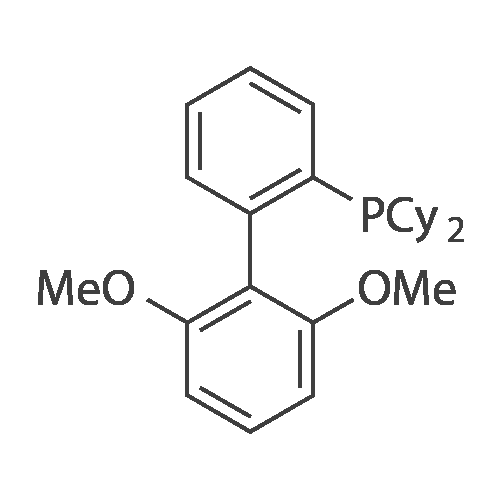
3-chlorobenzofuro[3,2-c]pyridine (2.89 g, 14 mmol), phenylboronic acid (2.56 g, 21 mmol), potassium phosphate (9.6 g, 42 mmol), 2-dicyclohexylphosphino-2',6'-dimethoxybiphenyl (0.45 g, 1.12 mmol) and Pd2(bda)3 (0.256 g, 0.28 mmol) were to toluene (100 mL) and water (10 mL). Nitrogen was bubbled through the solution for 30 minutes and then the solution was refluxed for overnight in an atmosphere of nitrogen. The reaction was then allowed to cool to room temperature and the organic phase was separated from the aqueous phase. The aqueous phase was washed with ethyl acetate and the organic fractions were combined and dried over sodium sulfate and the solvent removed under vacuum. The product was chromatographed using silica gel with ethyl acetate and hexanes as the eluent. The solvent was removed to give 2.77 g of title compound. Synthesis of Compound 1: Iridium intermediate (2.67 g, 3.76 mmol) and 3-phenylbenzofuro[3,2-c]pyridine (2.77 g, 11.29 mmol) was mixed in 50 mL of anhydrous ethanol. |
- 3
-
 [ 800402-12-4 ]
[ 800402-12-4 ]

-
 [ 1251041-55-0 ]
[ 1251041-55-0 ]
Categories

 Chemistry
Chemistry
 Pharmaceutical Intermediates
Pharmaceutical Intermediates
 Inhibitors/Agonists
Inhibitors/Agonists
 Material Science
Material Science














 For Research Only
For Research Only
 110K+ Compounds
110K+ Compounds
 Competitive Price
Competitive Price
 1-2 Day Shipping
1-2 Day Shipping








