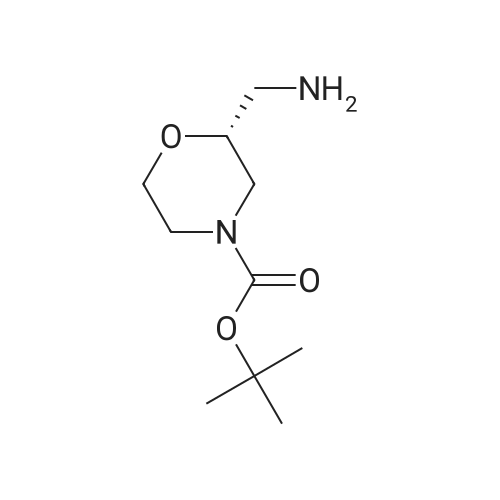
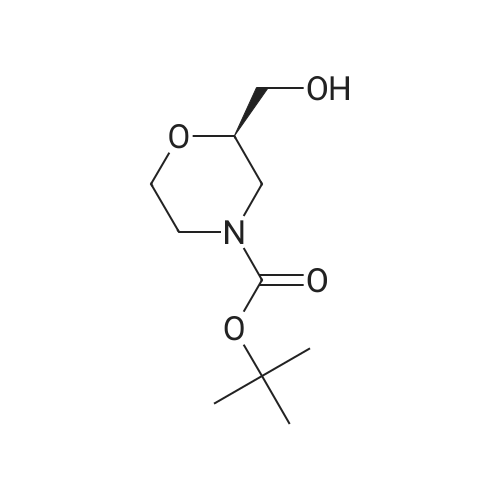
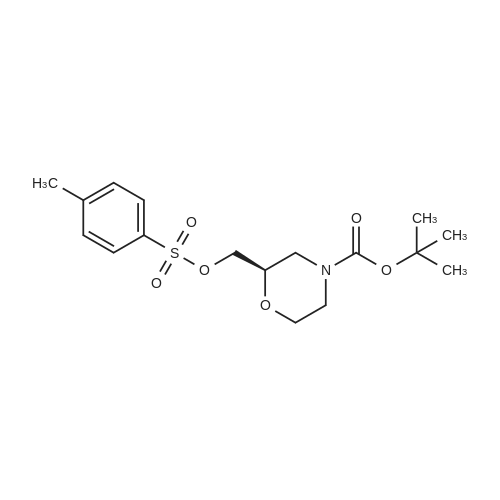
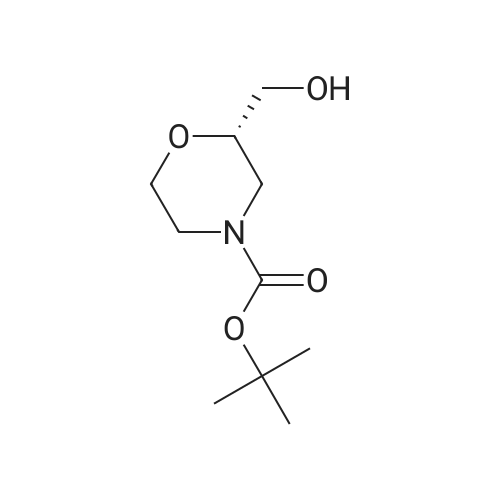
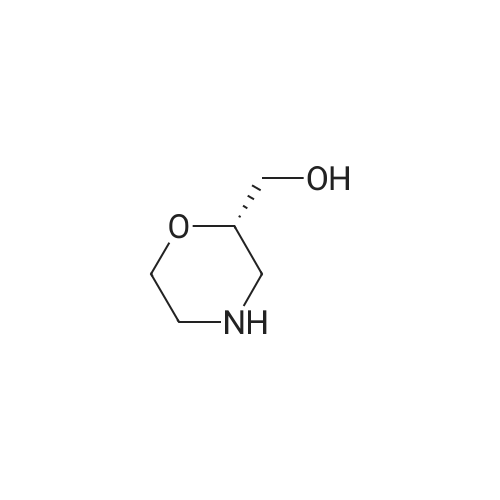
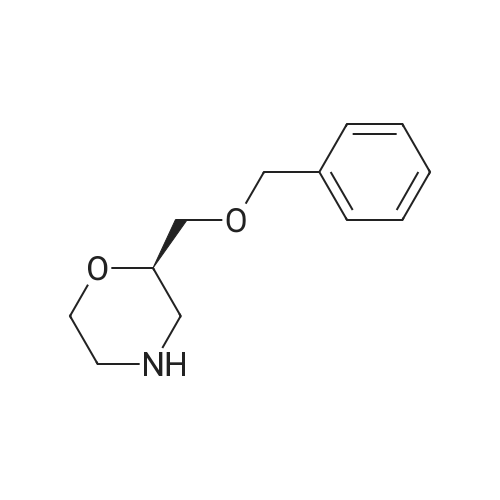
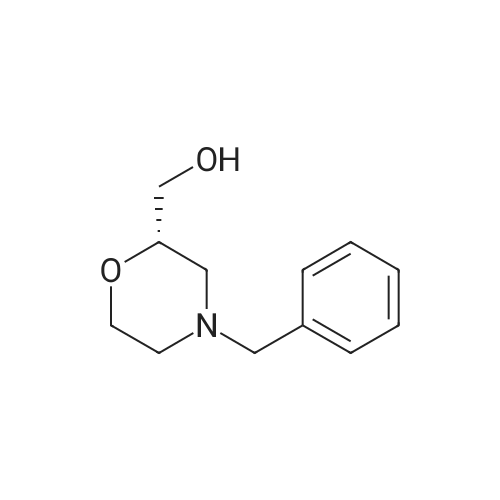
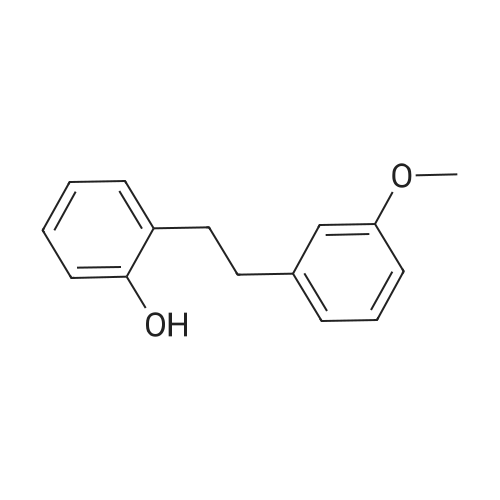
| 99% |
With trimethylamine hydrochloride; triethylamine; In dichloromethane; at 20℃; for 2h; |
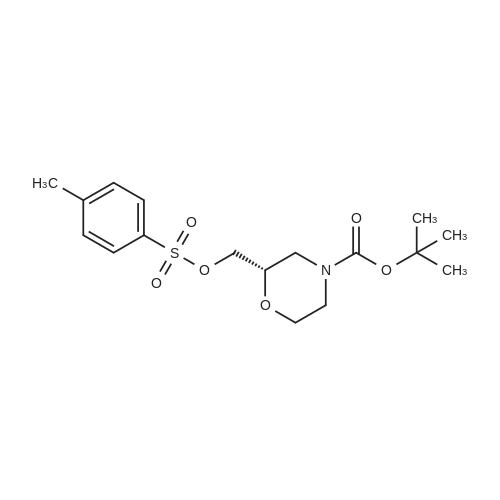
To a mixture of <strong>[135065-76-8]tert-butyl (2S)-2-(hydroxymethyl)morpholine-4-carboxylate</strong> (10.0 g, 46.0 mmol) (CAS-No.: 135065-76-8), triethylamine (9.6 ml, 69 mmol) and trimethylamine hydrochloride (440 mg, 4.6 mmol) in dichloromethane (120 ml) was added 4-methylbenzenesulfonyl chloride (13.2 g, 69.0 mmol). The reaction was stirred at rt for 2 h. After this time, the reaction mixture was treated with N,N-dimethylethane-1,2-diamine (3.0 ml, 27.6 mmol). The reaction mixture was washed with 1 M aqueous hydrogen chloride solution followed by water, dried (Na2S04), filtered and concentrated under reduced pressure to afford 18.3 g (99% yield) of the title compound as a yellow oil. 1H NMR (250 MHz, Chloroform-d) delta [ppm]: 1.45 (s, 9H), 2.45 (s, 3H), 2.56 - 2.75 (m, 1H), 2.77 - 2.97 (m, 1H), 3.46 (td, 1H), 3.52 - 3.67 (m, 1H), 3.72 - 3.92 (m, 3H), 3.93 - 4.10 (m, 2H), 7.35 (d, 2H), 7.80 (d, 2H). LC-MS (Analytical Method A) Rt = 1.23 min, MS (ESIpos): m/z = 272 [M-Boc+H]+. |
| 97% |
With triethylamine;dmap; In dichloromethane; for 24h; |
Toluene sulphonyl chloride (0.513 g, 2.69 mmol) was added to a solution of <strong>[135065-76-8](S)-tert-butyl 2-(hydroxymethyl)morpholine-4-carboxylate</strong> (0.390 g, 1.79 mmol), triethylamine (0.50 mL, 3.59 mmol) and DMAP (cat.) in dichloromethane (11 mL). The reaction mixture was <n="262"/>stirred for 24 h then diluted with dichloromethane (25 ml) and washed sequentially with water (25 ml_) and 0.2M HCI (25 mL). The organic phase was dried (Na2SO4) and the solvent was removed in vacuo. The crude product was purified by flash column chromatography on silica gel, eluting with ethyl acetate / hexane (1 / 5), to give the title compound as a colourless solid (0.650 g, 97%).1H NMR (CDCI3, 500 MHz1) delta 7.76 (d, 2H, J = 8.5 Hz), 7.31 (d, 2H, J = 8.5 Hz), 4.00-3.96 (m, 2H), 3.89-3.76 (m, 3H), 3.62-3.58 (m, 1 H), 3.48-3.43 (m, 1 H), 2.91-2.87 (m, 1 H), 2.69-2.65 (m, 1 H), 2.45 (s, 3H), 1.41 (s, 9H). LCMS (4) Rt = 2.53 min; m/z (ESI+) 394 [M+Na+]. |
| 55% |
With triethylamine; In dichloromethane; at 20℃;Cooling with ice; |
(S)-2-Hydroxymethyl-morpholine-4-carboxylic acid tert-butyl ester 4d (0.217 g, 1 mmol) was dissolved in 10 mL of dichloromethane with stirring, and added with p-toluenesulfonyl chloride (0.324 g, 1.7 mmol) and triethylamine (0.252 g, 2.5 mmol) in an ice-water bath. The reaction mixture was reacted overnight at room temperature and monitored by thin layer chromatography until the disappearance of the starting materials. The resulting mixture was quenched with saturated sodium bicarbonate solution (10 mL), and extracted with dichloromethane (50 mL×3). The combined organic phase was washed successively with water (50 mL) and saturated brine (50 mL), dried over anhydrous magnesium sulfate, filtered and concentrated under reduced pressure. The residue was purified by silica gel column chromatography to obtain the title compound (S)-2-(toluene-4-sulfonyloxymethyl)-morpholine-4-carboxylic acid tert-butyl ester 4e (0.203 g, yield 55%) as a colorless viscous liquid. MS m/z (ESI): 394.3 [M+23]. 1H NMR (CDCl3, 400 MHz) delta 7.8-7.78 (d, 2H), 7.36-7.34 (d, 2H), 4.05-3.97 (m, 2H), 3.9-3.8 (m, 3H), 3.61-3.58 (m, 1H), 3.49-3.43 (m, 1H), 2.9-2.85 (m, 1H), 2.7-2.61 (m, 1H), 2.452 (s, 3H), 1.45 (s, 9H). |
| 55% |
With triethylamine; In dichloromethane; at 20℃;Cooling with ice; |
(S)-2-Hydroxymethyl-morpholine-4-carboxylic acid tert-butyl ester 4d (0.217 g, 1 mmol) was dissolved in 10 mL of dichloromethane with stirring, and added with p-toluenesulfonyl chloride (0.324 g, 1.7 mmol) and triethylamine (0.252 g, 2.5 mmol) in an ice-water bath. The reaction mixture was reacted overnight at room temperature and monitored by thin layer chromatography until the disappearance of the starting materials. The resulting mixture was quenched with saturated sodium bicarbonate solution (10 mL), and extracted with dichloromethane (50 mL×3). The combined organic phase was washed successively with water (50 mL) and saturated brine (50 mL), dried over anhydrous magnesium sulfate, filtered and concentrated under reduced pressure. The residue was purified by silica gel column chromatography to obtain the title compound (S)-2-(toluene-4-sulfonyloxymethyl)-morpholine-4-carboxylic acid tert-butyl ester 4e (0.203 g, yield 55%) as a colorless viscous liquid.MS m/z (ESI): 394.3 [M+23].1H NMR (CDCl3, 400 MHz) delta 7.8-7.78 (d, 2H), 7.36-7.34 (d, 2H), 4.05-3.97 (m, 2H), 3.9-3.8 (m, 3H), 3.61-3.58 (m, 1H), 3.49-3.43 (m, 1H), 2.9-2.85 (m, 1H), 2.7-2.61 (m, 1H), 2.452 (s, 3H), 1.45 (s, 9H). |
|
With triethylamine; In dichloromethane; at 20℃; for 72h;Inert atmosphere; |
(0763) To a solution of <strong>[135065-76-8](S)-tert-butyl 2-(hydroxymethyl)morpholine-4-carboxylate</strong> (1 g) in dichloromethane (50 ml) was added triethylamine (1.604 ml) and 4-methylbenzene-1-sulfonyl chloride (1.097 g). The mixture was stirred at ambient temperature under nitrogen for 72 hours. The reaction was diluted with methylene chloride (50 ml) and brine (100 ml). (0764) The brine layer was extracted with methylene chloride (75 ml). The combined organics were dried over sodium sulfate, filtered and concentrated. The crude material was purified on a silica gel column eluting with a 15-65% ethyl acetate in hexane gradient to provide the title compound. |
|
With dmap; triethylamine; In dichloromethane; at 25℃; for 16h; |
To a solution of (5)-/er/-butyl 2-(hydroxymethyl)morpholine-4-carboxylate (1.0 g, 4.6 mmol, compound C8.1) in DCM (5 mL) was added p-toluenesulfonyl chloride (0.97 g, 5.06 mmol), triethylamine (0.64 mL, 4.6 mmol) and 4-dimethylaminopyridine (56.23 mg, 0.460 mmol) and the resulting reaction mixture was stirred at 25 C for 16 h. The mixture was diluted with 20 mL EtOAc and filtered, the filtrate was then concentrated in vacuo to give a crude product of {S)-tert- butyl 2-((tosyloxy)methyl)morpholine-4-carboxylate (2 g, 5.38 mmol, 95.92% yield). MS (ESI) : 394.2 ([M+Na]+). It was used directly in the next step without further purification. |

 Chemistry
Chemistry
 Pharmaceutical Intermediates
Pharmaceutical Intermediates
 Inhibitors/Agonists
Inhibitors/Agonists
 Material Science
Material Science














 For Research Only
For Research Only
 110K+ Compounds
110K+ Compounds
 Competitive Price
Competitive Price
 1-2 Day Shipping
1-2 Day Shipping