Alternatived Products of [ 153903-21-0 ]
Product Details of [ 153903-21-0 ]
| CAS No. : | 153903-21-0 |
MDL No. : | |
| Formula : |
C12H16FNO2
|
Boiling Point : |
- |
| Linear Structure Formula : | - |
InChI Key : | - |
| M.W : |
225.26
|
Pubchem ID : | - |
| Synonyms : |
|
Safety of [ 153903-21-0 ]
| Signal Word: | |
Class: | |
| Precautionary Statements: | |
UN#: | |
| Hazard Statements: | |
Packing Group: | |
Application In Synthesis of [ 153903-21-0 ]
* All experimental methods are cited from the reference, please refer to the original source for details. We do not guarantee the accuracy of the content in the reference.
- Upstream synthesis route of [ 153903-21-0 ]
- Downstream synthetic route of [ 153903-21-0 ]
- 1
-
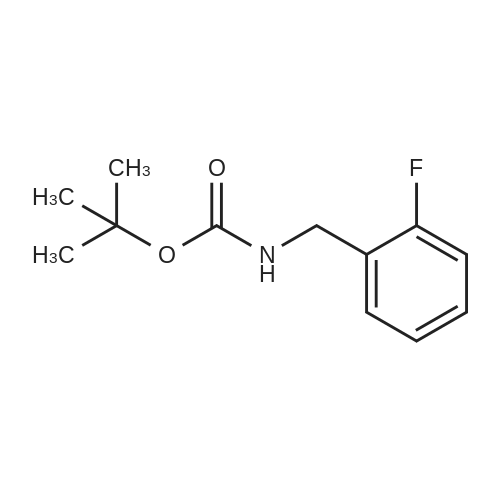 [ 153903-21-0 ]
[ 153903-21-0 ]

-
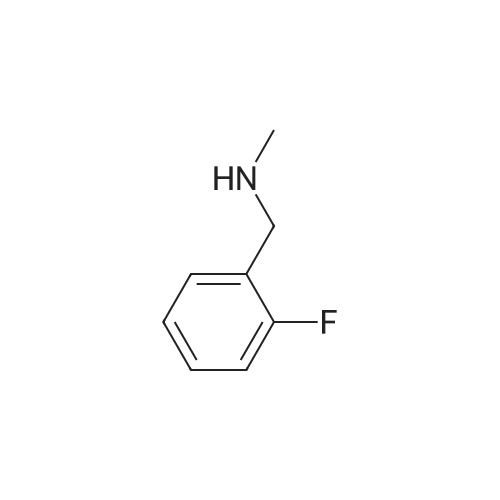 [ 399-30-4 ]
[ 399-30-4 ]
| Yield | Reaction Conditions | Operation in experiment |
|
|
PREPARATIONS; Example 1; Procedure for making an intermediate according to Scheme A. F F 1. Boc20, Et3N, DCM, rt H2N Y HN 2. LiAIH4, THF, reflux (2-Fluoro-benzyl)-methyl amine; To a solution of 300 mg (2.40 mmol) of 2-fluorobenzylamine, 29.3 mg (0.24 mmol) of 4- dimethylaminopyridine and 342 ul (2. 64 mmol) of triethylamine in 3 mL of dichloromethane is added 628 mg (2.88 mmol) of di (tert-butoxycarbonyl) in 3 mL of dichloromethane at 0 C. The reaction is allowed to react for 72 h and solvents are removed under reduced pressure. The crude product is dissolved in 5 mL of 1N hydrochloric acid solution and extracted three times with ethyl acetate. The organic phase is separated and washed with saturated sodium bicarbonate solution and brine and dried with sodium sulphate. Removal of the solvent affords (2-fluoro-benzyl)- carbamic acid tert-butyl ester, which is taken directly to the next step. To a solution of 250 mg (1.11 mmol) of the carbamic ester dissolved in 1 mL of tetrahydrofuran is added 1.66 mL of a 1 M lithium aluminiumhydride solution in tetrahydrofuran. The reaction mixture is stirred at room temperature until gas evolution has ceased and is further heated at reflux temperature for 3 h. After cooling to room temperature, 1N hydrochloric acid solution is added and the aqueous phase is extracted with dichloromethane. The aqueous phase is separated, a saturated sodium bicarbonate solution is added until pH reaches 7-8 and dichloromethane is added. The organic layer is extracted with brine and water, dried with sodium sulphate and concentrated under vacuum to obtain the title compound. LC/MS (III) rt 1.52, m/z 140 [M+H]+. |
- 2
-
 [ 124-38-9 ]
[ 124-38-9 ]

-
 [ 1174761-19-3 ]
[ 1174761-19-3 ]

-
 [ 153903-21-0 ]
[ 153903-21-0 ]

-
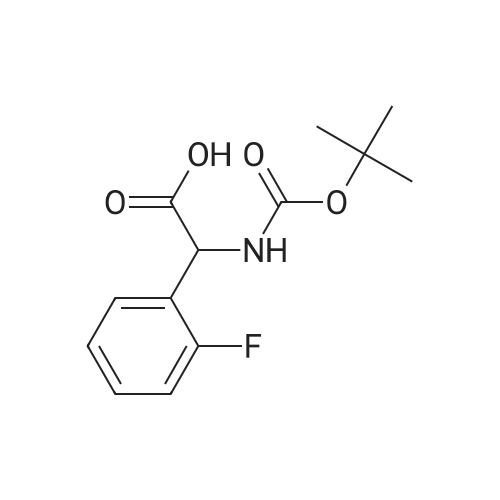 [ 161330-30-9 ]
[ 161330-30-9 ]
| Yield | Reaction Conditions | Operation in experiment |
|
Stage #1: N-(tert-butoxycarbonyl)-α-(phenylsulfonyl)-2-fluorobenzylamine With trimethylsilyltributyltin; cesium fluoride In N,N-dimethyl-formamide at 20℃; for 0.0833333h; Inert atmosphere;
Stage #2: carbon dioxide In N,N-dimethyl-formamide at 100℃; for 3h; Autoclave;
Stage #3: With hydrogenchloride In diethyl ether; water; N,N-dimethyl-formamide Inert atmosphere; |
|
- 3
-
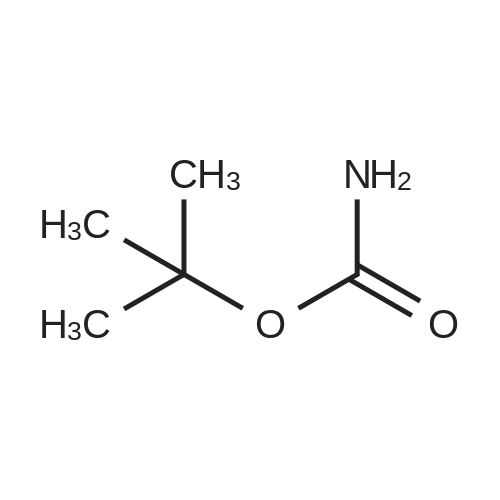 [ 4248-19-5 ]
[ 4248-19-5 ]

-
 [ 153903-21-0 ]
[ 153903-21-0 ]

-
 [ 161330-30-9 ]
[ 161330-30-9 ]
| Yield | Reaction Conditions | Operation in experiment |
|
Multi-step reaction with 2 steps
1.1: formic acid / methanol; water / 60 h / 20 °C / Inert atmosphere
2.1: trimethylsilyltributyltin; cesium fluoride / N,N-dimethyl-formamide / 0.08 h / 20 °C / Inert atmosphere
2.2: 3 h / 100 °C / 7500.75 Torr / Autoclave
2.3: pH 2 / Inert atmosphere |
|
- 4
-
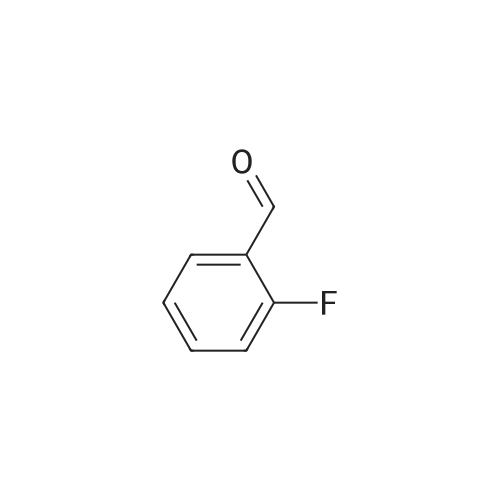 [ 446-52-6 ]
[ 446-52-6 ]

-
 [ 153903-21-0 ]
[ 153903-21-0 ]

-
 [ 161330-30-9 ]
[ 161330-30-9 ]
| Yield | Reaction Conditions | Operation in experiment |
|
Multi-step reaction with 2 steps
1.1: formic acid / methanol; water / 60 h / 20 °C / Inert atmosphere
2.1: trimethylsilyltributyltin; cesium fluoride / N,N-dimethyl-formamide / 0.08 h / 20 °C / Inert atmosphere
2.2: 3 h / 100 °C / 7500.75 Torr / Autoclave
2.3: pH 2 / Inert atmosphere |
|
- 5
-
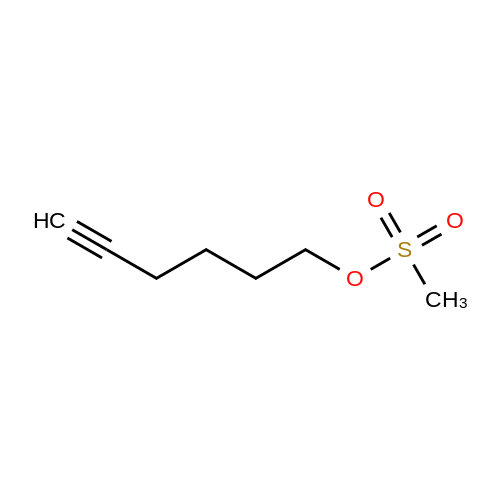 [ 79496-61-0 ]
[ 79496-61-0 ]

-
 [ 153903-21-0 ]
[ 153903-21-0 ]

-
tert-butyl 2-fluorobenzyl(hex-5-yn-1-yl)carbamate
[ No CAS ]
| Yield | Reaction Conditions | Operation in experiment |
| 70% |
With sodium hydride; In N,N-dimethyl-formamide; at 20℃; for 64h; |
General procedure: To a suspension of NaH (60% dispersion,1.04 g, 26 mmol) in DMF (80 mL) was added a solution of 12a (4.51 g, 20 mmol) in DMF (10 mL) at 0 C. After stirring at room temperature for 4 h, a solution of 10a (5.6 g, 22 mmol) in DMF (10 mL) was added, and the mixture was stirred for 59 h. Satd. NH4Cl aq (40 mL) was added, and the whole was extracted with Et2O (200 + 100 + 100 mL). The combined organic layers were washed with water (500 + 200 mL), brine (200 mL), dried over Na2SO4 and concentrated. Column chromatography (SiO2 350 g, hexane/EtOAc 10/1 to 2/1) gave 13a (4.95 g, 65%, E/Z 9/1) as a pale yellow oil. |

 Chemistry
Chemistry
 Pharmaceutical Intermediates
Pharmaceutical Intermediates
 Inhibitors/Agonists
Inhibitors/Agonists
 Material Science
Material Science














 For Research Only
For Research Only
 110K+ Compounds
110K+ Compounds
 Competitive Price
Competitive Price
 1-2 Day Shipping
1-2 Day Shipping









