Alternatived Products of [ 17626-02-7 ]
Product Details of [ 17626-02-7 ]
| CAS No. : | 17626-02-7 |
MDL No. : | MFCD00465199 |
| Formula : |
C8H12N2O2
|
Boiling Point : |
- |
| Linear Structure Formula : | - |
InChI Key : | - |
| M.W : |
168.19
|
Pubchem ID : | - |
| Synonyms : |
|
Application In Synthesis of [ 17626-02-7 ]
* All experimental methods are cited from the reference, please refer to the original source for details. We do not guarantee the accuracy of the content in the reference.
- Downstream synthetic route of [ 17626-02-7 ]
- 1
-
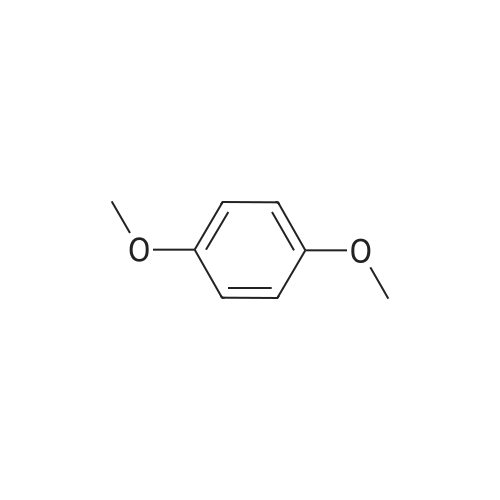 [ 150-78-7 ]
[ 150-78-7 ]

-
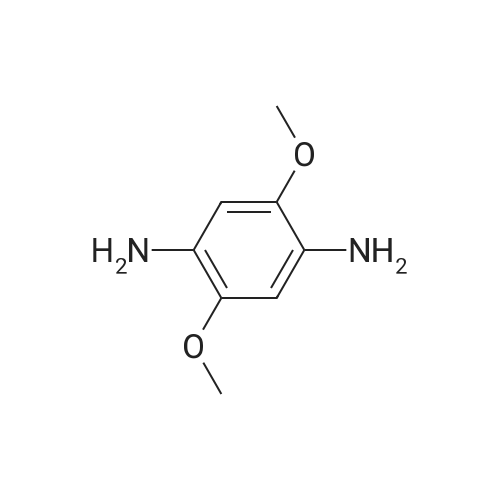 [ 17626-02-7 ]
[ 17626-02-7 ]

-
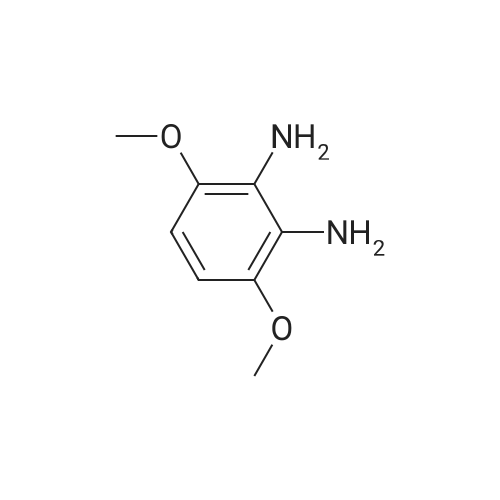 [ 40328-95-8 ]
[ 40328-95-8 ]
| Yield | Reaction Conditions | Operation in experiment |
|
With sodium dithionite; nitric acid 1.) glacial acetic acid, 80-90 deg C, 5 min, 2.) THF, MeOH, reflux, 1 h; Yield given. Multistep reaction. Yields of byproduct given; |
|
|
Stage #1: 1,4-dimethoxybezene With nitric acid In acetic acid
Stage #2: With hydrogen In ethanol |
|
|
Stage #1: 1,4-dimethoxybezene With nitric acid In acetic acid
Stage #2: With palladium 10% on activated carbon; hydrogen In methanol at 20℃; for 24h; |
|
|
Stage #1: 1,4-dimethoxybezene With nitric acid; acetic acid for 0.0833333h;
Stage #2: With palladium 10% on activated carbon; hydrogen In ethyl acetate for 24h; Autoclave; Overall yield = 5.84 g; |
|

Reference:
[1]Shaikh; Johnson; Grollman
[Journal of Medicinal Chemistry, 1986, vol. 29, # 8, p. 1329 - 1340]
[2]Lion, Claude; Baudry, Richard; Hedayatullah, Mir; Da Conceicao, Louis; Genard, Sylvie; Maignan, Jean
[Journal of Heterocyclic Chemistry, 2002, vol. 39, # 1, p. 125 - 130]
[3]Location in patent: experimental part
Shen, De-Qing; Wu, Ning; Li, Yan-Ping; Wu, Zu-Ping; Zhang, Hong-Bin; Huang, Zhi-Shu; Gu, Lian-Quan; An, Lin-Kun
[Australian Journal of Chemistry, 2010, vol. 63, # 7, p. 1116 - 1121]
[4]Kolmer-Anderl, Nicole; Kolmer, Andreas; Thiele, Christina M.; Rehahn, Matthias
[Chemistry - A European Journal, 2016, vol. 22, # 15, p. 5277 - 5287]
- 2
-
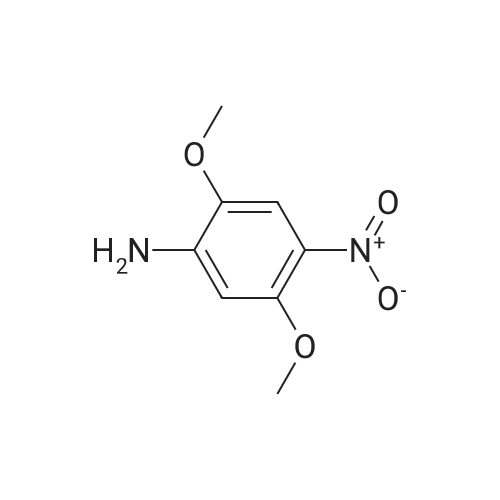 [ 6313-37-7 ]
[ 6313-37-7 ]

-
 [ 17626-02-7 ]
[ 17626-02-7 ]
| Yield | Reaction Conditions | Operation in experiment |
| 88% |
With hydrogen In ethanol for 15h; |
|
|
With palladium 10% on activated carbon; hydrogen In ethanol at 20℃; for 16h; |
5
To a solution of compound 5.1 (1.00 g, 5.05 mmol, 1 equiv) in EtOH (150 mL) was added Pd/C (0.30 g, 10.0% purity) in one portion, then the reaction mixture was stirred at 20 °C for 16 hr under a Eh atmosphere (15 psi). TLC (dichloromethane/MeOH = 15/1) showed compound 5.1 (Rf = 0.70) was consumed. A new spot could be observed (Rf = 0.41). The reaction mixture was filtered through a celite pad, and the filtrate was concentrated to give compound 5.2 (800 mg, crude) as a black-brown solid. |
Reference:
[1]Miller, Scott E.; Lukas, Aaron S.; Marsh, Emily; Bushard, Patrick; Wasielewski, Michael R.
[Journal of the American Chemical Society, 2000, vol. 122, # 32, p. 7802 - 7810]
[2]Current Patent Assignee: FOG PHARMACEUTICALS - WO2020/41270, 2020, A1
Location in patent: Paragraph 0360-0361
- 3
-
 [ 56741-26-5 ]
[ 56741-26-5 ]

-
 [ 17626-02-7 ]
[ 17626-02-7 ]
| Yield | Reaction Conditions | Operation in experiment |
| 99% |
With hydrogen; palladium(II) hydroxide In methanol; ethyl acetate at 25℃; for 6h; |
|
|
With hydrogen In tetrahydrofuran at 20℃; for 7h; |
|
Reference:
[1]Wang, Christy; Sperry, Jonathan
[Tetrahedron, 2013, vol. 69, # 23, p. 4563 - 4577]
[2]Wu, Jing; Fang, Fang; Lu, Wen-Ya; Hou, Jun-Li; Li, Chuang; Wu, Zong-Quan; Jiang, Xi-Kui; Li, Zhan-Ting; Yu, Yi-Hua
[Journal of Organic Chemistry, 2007, vol. 72, # 8, p. 2897 - 2905]

 Chemistry
Chemistry
 Pharmaceutical Intermediates
Pharmaceutical Intermediates
 Inhibitors/Agonists
Inhibitors/Agonists
 Material Science
Material Science















 For Research Only
For Research Only
 110K+ Compounds
110K+ Compounds
 Competitive Price
Competitive Price
 1-2 Day Shipping
1-2 Day Shipping









