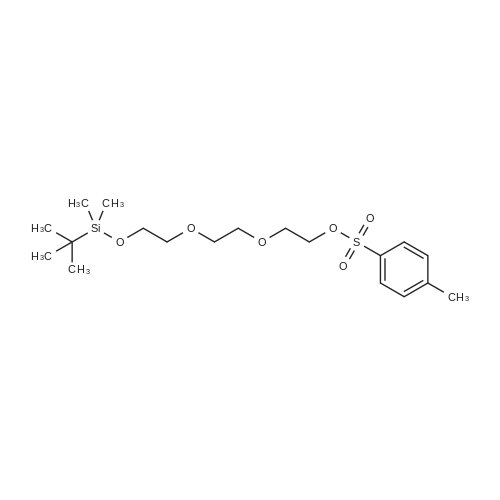
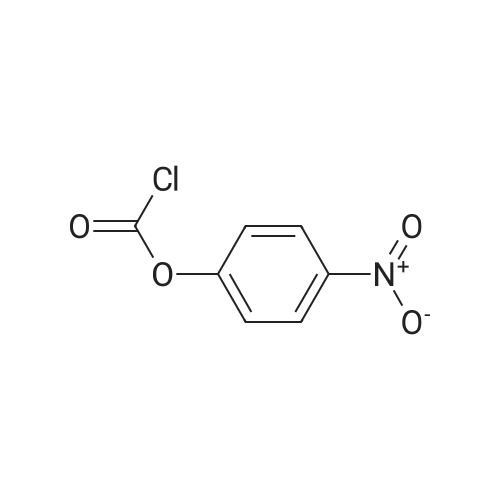
| 80.2% |
With dmap; triethylamine In dichloromethane at 0 - 20℃; for 4h; |
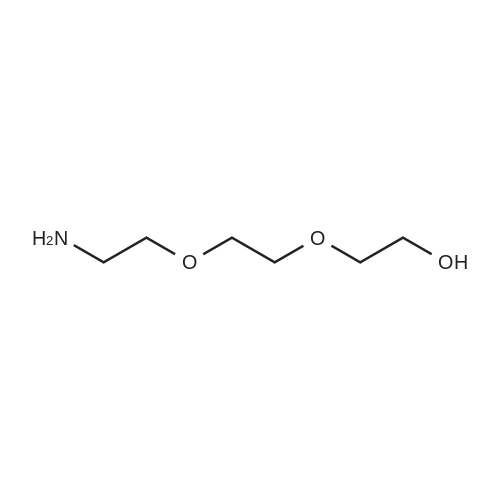
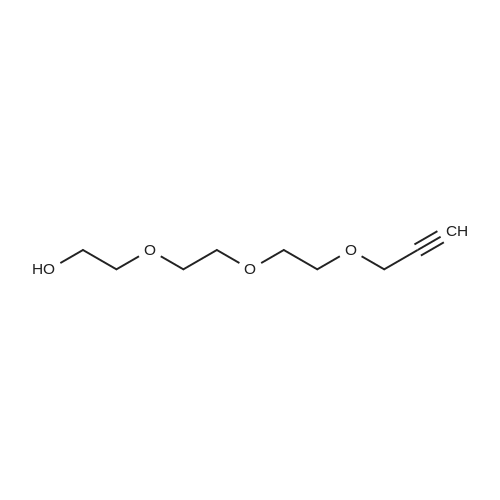
29 [00161] Example 29: Synthesis of N-methyl-2-((1-(2-(2-(2- hydrosxyethoxy)ethoxy)ethoxcarbonyl) -3-((1E)-2-(2-pyridinyl)ethenyl)-1H-indazol-6- yl)thio)benzamide hydrochloride (compound 50).
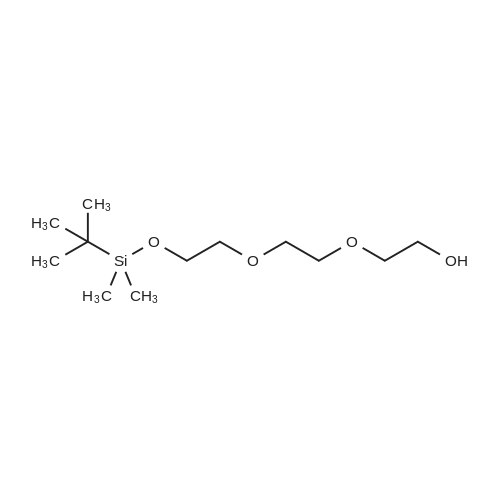
[00162] In a flask were added triethylene glycol (5.0 g, 33.0 mmol, 5.0 eq.), DCM (10 mL), DMAP (0.16 g, 1.3 mmol, 0.2 eq.), TEA (0.67 g, 6.6 mmol, 1.0 eq.), and DCM (10 mL). The mixture was cooled to 0 °C, followed by dropwise addition of a solution of TBSC1 (1.0 g, 6.6 mmol, 1.0 eq.) in DCM (2.5 mL). The reaction temperature was raised to rt; and the mixture was stirred at rt for 4 h. The mixture was then washed with saturated ammonium chloride solution (30 mL) and with brine (30 mL), subsequently. The organic layer was concentrated to dryness, providing 10,10,11,11-tetramethyl-3, 6,9- trioxa-10-siladodecan-1-ol (1.4 g, 80.2%). |
| 73% |
With pyridine In dichloromethane at 20℃; |
|
| 64% |
With dmap; triethylamine In dichloromethane at 20℃; for 3h; Inert atmosphere; |
|
| 55% |
With sodium hydride In tetrahydrofuran at 20℃; for 1h; |
|
| 52% |
With 1H-imidazole In dichloromethane at 20℃; for 17h; |
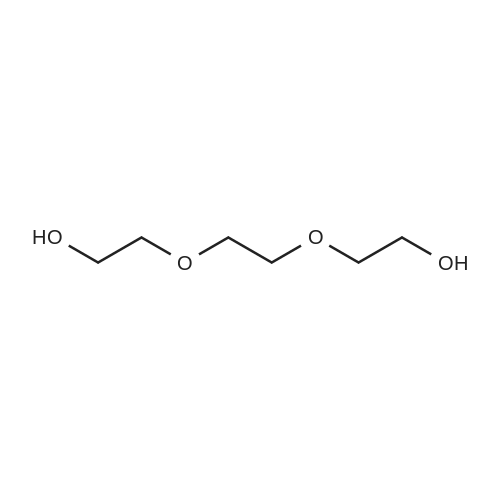
j00710j 2,2,3,3-Tetramethyl-4,7,1 0-trioxa-3-siladodecan-12-ol (2):
1007111 A solution of 2,2’-(ethane-1,2-diylbis(oxy))bis(ethan-1-ol) (15 g, 99.5 mmol) inDCM (150 mL) at 0 °C was charged with imidazole (4.7 g, 69.6 mmol), TBDMS chloride (10.5 g, 69.6 mmol) and stirred at 0°C for 1 h. The resulting solution was warmed to room temperature and stirred for 16 h. The reaction mixture was partition between with H20 (100 mL) and DCM (100 mL) and the separated organic layer was washed with 10% aqueous HC1 solution, saturated sodium bicarbonate and brine. The combined organic layers were dried overanhydrous Na2SO4, filtered and concentrated in vacuo resulting in a crude compound which was purified by chromatography on silica gel, eluting with 2-4% methanol in DCMto gvie 13.8 g, 52% yield of the title compound as a light yellow oil. ‘H NMR (400 MHz, DMSO-d6): ö = 5.22 (s, 1H), 4.51 (t, J= 5.51 Hz, 2H), 3.64 (d, J= 4.41 Hz, 4H), 3.43 -3.53 (m, 6H), 0.82 (s, 9H), 0.08 (s, 6H). |
| 51% |
With dmap; triethylamine In dichloromethane at 0 - 25℃; Inert atmosphere; |
|
| 50% |
With triethylamine In dichloromethane |
|
| 48% |
With 1H-imidazole In N,N-dimethyl-formamide at 0 - 20℃; for 16h; |
|
| 28% |
With 1H-imidazole In dichloromethane at 0 - 20℃; for 10h; |
23.A1 2,2,3,3-tetramethyl-4,7,10-trioxa-3-siladodecan-12-ol (23-2B 1):
To a stirred solution of 23-1 (20 g, 133.33 mmol) in dichlorome thane (100 mL) was added imidazole (5.4 g, 79.99 mmol) and TBDMSC1 (10.0 g g, 66.66 mmol) at 0 °C and stirred to RT for 10 hr. The reaction mixture was diluted with DCM (200 mL) and washed with water (2 X 100 mL) and brine (50 mL). The organic layer was concentrated under reduced pressure and the crude residue was purified by flash column chromatography (100-200 silica) using 20% ethyl acetate/pet. ether gave 23-2B1 (10 g, 37.87 mmol, 28% yield) as a colorless liquid. |
| 17% |
With triethylamine In hexane; acetonitrile at 20℃; for 24h; Inert atmosphere; |
Typical procedure for the monosilylation of symmetric diols
General procedure: In a flame-dried100 mL round-bottom flask equipped with a teflon magnetic stir bar,diol(5.0 mmol) was added to acetonitrile (12.5 mL) and hexanes (37.5 mL).Triethylamine (Et3N, 0.84 mL, 6.00 mmol, 1.2 equiv) and tertbutyldimethylsilylchloride (TBSCl, 0.53 g, 5.0 mmol, 1.0 equiv) were addedto the solution and the resulting biphasic mixture was stirred vigorously atroom temperature for 24 h under a N2 atmosphere. The reaction was quenchedwith saturated aqueous ammonium chloride (NH4Cl, 50 mL), and extractedwith ethyl acetate (EtOAc, 3 50 mL). The combined organic phase waswashed with brine (3 50 mL) and dried over anhydrous sodium sulfate(Na2SO4). The crude product was purified by silica gel column chromatography(10-15% EtOAc/hexanes). |

 Chemistry
Chemistry
 Pharmaceutical Intermediates
Pharmaceutical Intermediates
 Inhibitors/Agonists
Inhibitors/Agonists
 Material Science
Material Science














 For Research Only
For Research Only
 110K+ Compounds
110K+ Compounds
 Competitive Price
Competitive Price
 1-2 Day Shipping
1-2 Day Shipping