|
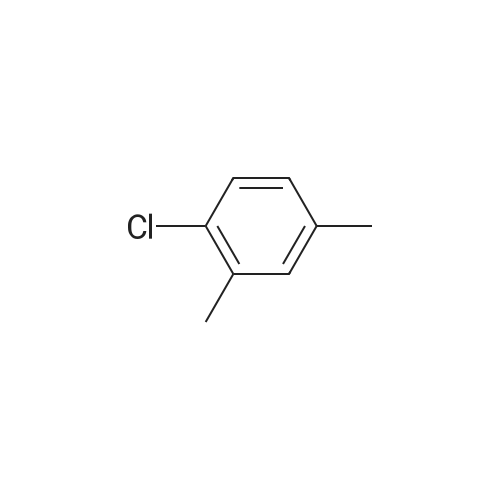
With chlorine;the fluorine-containing K-L-type zeolite; In 1,2-dichloro-ethane; at 80℃; for 1.75 - 3.66667h;Product distribution / selectivity; |
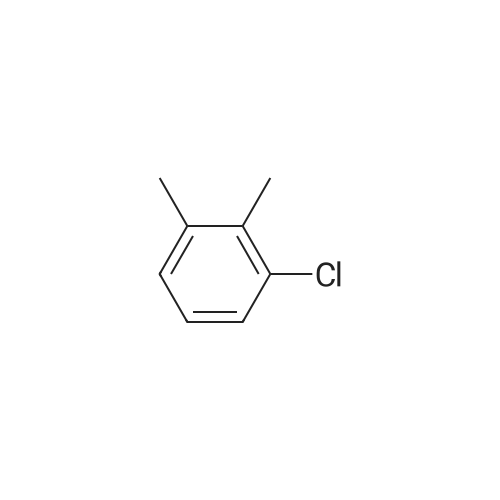
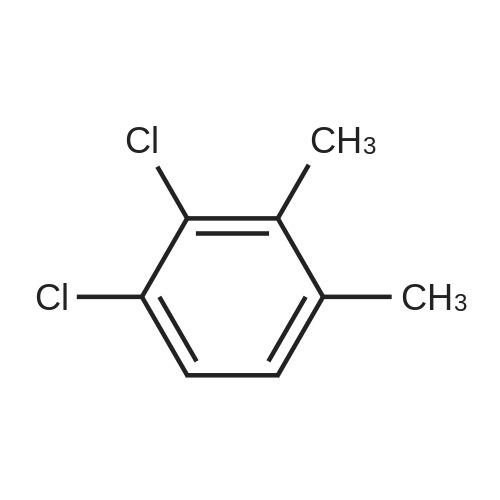
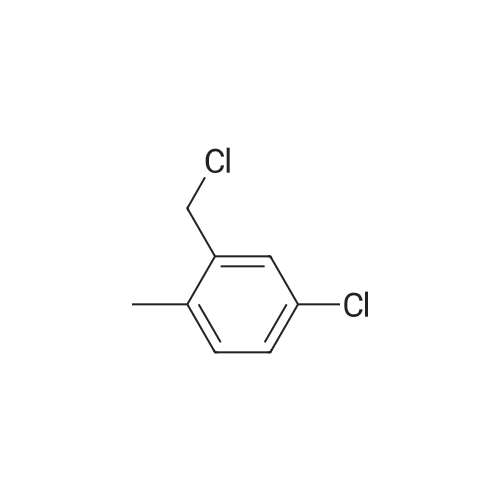
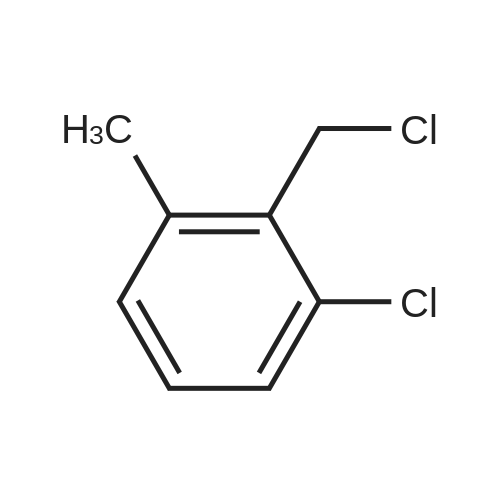
A 200-ml four-neck flask was equipped with a reflux condenser, a thermometer, a chlorine inlet tube, and a sampling duct. This was so designed that waste chlorine gas (hydrochloric acid gas) could be exhausted from the top of the reflux condenser and trapped in an aqueous sodium hydroxide solution. Heating it was effected in an oil bath. 17.1 g of orthoxylene orthoxylene (Sigma Aldrich Japan's first class grade chemical) that had been previously dried with Molecular Sieve 4A to have a water content of 20 ppm, 74.8 g of 1, 2-dichloroethane (Sigma Aldrich Japan's first class grade chemical), and 3.43 g of the fluorine-containing K-L-type zeolite (from Tosoh) that had been prepared in Example 1 and calcinated at 400C for 1 hour and then cooled in a desiccator were put into the flask, and heated at 80C with nitrogen gas being introduced thereinto through the chlorine gas inlet tube. After this was heated at 80C, the nitrogen gas was exchanged for chlorine gas and the reaction was started. The chlorine gas flow rate was controlled to be about 0.03 mol/hr. After 3 hours and 40 minutes, the chlorine gas introduction was stopped, the flask was bubbled with nitrogen for 1 hour, then the reaction solution was cooled to room temperature, and the catalyst was taken out through filtration. The reaction solution (filtrate) was concentrated, and 31 g of a crude product having a composition ratio shown in Table 1 and Table 2 was obtained.; (Reference Example 3) Preparation of mixture of chloroalkylbenzene derivative with an impurity of alpha-chloroalkylbenzene (benzyl chloride) derivative: A 1000-ml four-neck flask was equipped with a reflux condenser, a thermometer, a chlorine inlet tube, and a sampling duct. This was so designed that waste chlorine gas could be exhausted from the top of the reflux condenser and trapped in an aqueous sodium hydroxide solution. Heating it was effected in an oil bath. 89.18 g of orthoxylene (Sigma Aldrich Japan' s first class grade chemical) that had been previously dried with Molecular Sieve 4A to have a water content of 20 ppm, 315 ml of 1,2-dichloroethane (Sigma Aldrich Japan's first class grade chemical), and 17 . 8 g of the fluorine-containing K-L-type zeolite (from Tosoh) that had been prepared in Example 1 and calcinated at 400C for 1 hour and then cooled in a desiccator were put into the flask, and heated at 80C with nitrogen gas being introduced thereinto through the chlorine gas inlet tube. After this was heated at 80C, the nitrogen gas was exchanged for chlorine gas and the reaction was started. The chlorine gas flow rate was controlled to be about 0.82 mol/hr. After 1 hour and 45 minutes, the chlorine gas introduction was stopped, the flask was bubbled with nitrogen for 1 hour, then the reaction solution was cooled to room temperature, and the catalyst was taken out through filtration. The reaction solution (filtrate) was concentrated, and 144 g of a product having a composition ratio shown in Table 1 and Table 2 was obtained. |
|
With chlorine;a K-L-type zeolite; In 1,4-dioxane; 1,2-dichloro-ethane; at 80℃; for 6.75h; |
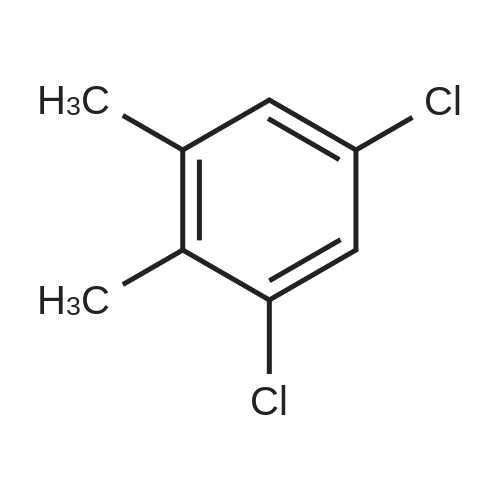
A 100-ml four-neck flask was equipped with a reflux condenser, a thermometer, a chlorine inlet tube, and a sampling duct. This was so designed that waste chlorine gas could be exhausted from the top of the reflux condenser and trapped in an aqueous sodium hydroxide solution. Heating it was effected in an oil bath. 12.7 g of orthoxylene (Sigma Aldrich Japan's first class grade chemical) that had been previously dried with Molecular Sieve 4A to have a water content of 20 ppm, 30 ml of 1,2-dichloroethane (Sigma Aldrich Japan's first class grade chemical), 0.56 g of 1,4-dioxane (Katayama Chemical Industry's special grade chemical), and 1.69 g of a K-L-type zeolite (from Tosoh) that had been calcinated at 400C for 1 hour and then cooled in a desiccator were put into the flask, and heated at 80C with nitrogen gas being introduced thereinto through the chlorine gas inlet tube. After this was heated at 80C, the nitrogen gas was exchanged for chlorine gas and the reaction was started. The chlorine gas flow rate was controlled to be about 0.04 mol/hr. After 6 hours and 45 minutes , the chlorine gas introduction was stopped, the flask was bubbled with nitrogen for 1 hour, then the reaction solution was cooled to room temperature, and the catalyst was taken out through filtration. The reaction solution (filtrate) was concentrated, and 23 g of a product having a composition ratio shown in Table 1 and Table 2 was obtained. |

 Chemistry
Chemistry
 Pharmaceutical Intermediates
Pharmaceutical Intermediates
 Inhibitors/Agonists
Inhibitors/Agonists
 Material Science
Material Science














 For Research Only
For Research Only
 110K+ Compounds
110K+ Compounds
 Competitive Price
Competitive Price
 1-2 Day Shipping
1-2 Day Shipping