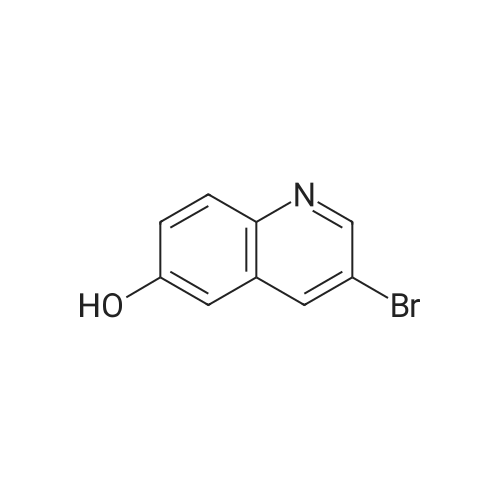
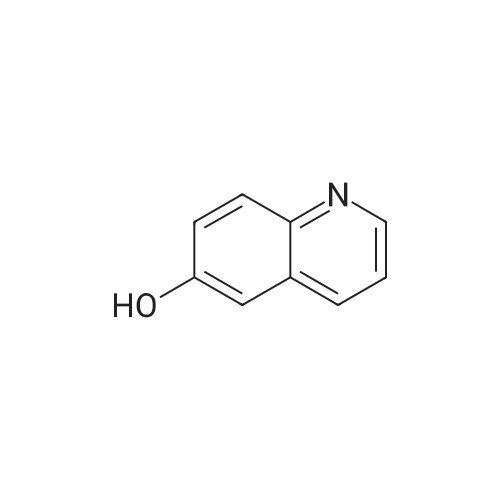
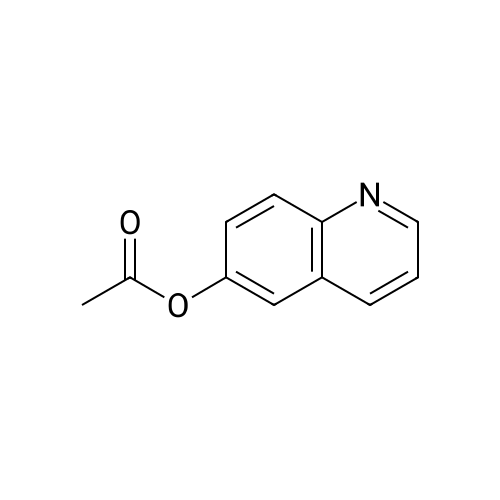
| 86% |
With pyridine In dichloromethane at 0 - 25℃; for 8h; Inert atmosphere; |
194 Example 194: Synthesis of quinolin-6-yl acetate (196
To a solution of 194 (24.0 g, 165 mmol) and pyridine (15.7 g, 198 mmol) in DCM (300 mL) was added 195 (15.5 g, 198 mmol) dropwise at 0° C. Then the mixture was stirred at 25° C. for 8 h under N2. The mixture was basified by sat. NaHCO3 solution to pH 8 and extracted by EA (2×300 mL). The organic layer was concentrated in vacuum and purified by silica gel column (0 to 36% of EA in PE) to obtain 196 (26.8 g, 86% yield, 99% purity) as yellow solid. [M+H]+ calcd for C11H9NO2 188.06, found 188.0. |
| 86% |
With pyridine In dichloromethane at 0 - 25℃; for 8h; Inert atmosphere; |
49.A Step A: Preparation of quinolin-6-yl acetate (49-2).
To a solution of 49-1 (24.0 g, 165 mmol) and pyridine (15.7 g, 198 mmol) in DCM (300 mL) was added acetyl chloride (15.5 g, 198 mmol) dropwise at 0 °C. Then the mixture was stirred at 25 °C for 8 h under N2. The mixture was basified by sat. NaHCO3 solution to pH=8 and extracted by EA (300 mL*2). The organic layer was concentrated in vacuum and purified by silica gel column (0 to 36% of EA in PE) to obtain 49-2 (26.8 g, 86% yield, 99% purity) as yellow solid. [M+H]+ calcd for C11H9NO2 188.06, found 188.0. |
| 86% |
With pyridine In dichloromethane at 0 - 20℃; for 8h; |
|
| 69% |
With pyridine at 0 - 20℃; for 3h; |
1.17.1
Intermediate 17: Trifluoro-methanesulfonic acid 3-(l-methyl-l//-pyrazol-4-yl)- quinolin-6-yl ester; Step 1: Acetic acid quinolin-6-yl ester; [0330] Quinolin-6-ol (135 g, 0.93 mol) was dissolved in pyridine (500 mL) and cooled to O0C in an ice-bath under a flow of nitrogen. Acetyl chloride (79 mL, 1.1 6mol) was added to the reaction mixture slowly. Then it was stirred at room temperature for 3 hours. The mixture was partitioned between ethyl acetate (400 mL) and saturated aqueous NaHCO3 (200 mL). The organic phase was separated and washed with brine (5*200mL). The organic phase was dried over Na2SO4, filtered and concentrated in vacuo. The residue was purified by column chromatography to afford 12O g of acetic acid quinolin-6-yl ester as white solid (69 % yield). |
| 69% |
With pyridine at 0 - 20℃; for 3h; |
1
Quinolin-6-ol (135 g, 0.93 mol) was dissolved in pyridine (500 mL) and cooled to O0C in an ice-bath under a flow of nitrogen. Acetyl chloride (79 mL, 1.1 6mol) was added to the reaction mixture slowly. Then it was stirred at room temperature for 3 hours. The mixture was partitioned between ethyl acetate (400 mL) and saturated aqueous NaHCO3 (200 mL). The organic phase was separated and washed with brine (5 x 20OmL). The organic phase was dried over Na2SOzJ, filtered and concentrated in vacuo. The residue was purified by column chromatography to afford 120 g of acetic acid quinolin-6-yl ester as white solid (69 % yield). |
| 68.9% |
With pyridine In dichloromethane at 0 - 20℃; for 8h; |
To a solution of quinolin-6-ol (4.5 g, 31.0 mmol) and pyridine (3.01 ml, 37.2 mmol) in DCM (50 ml) was added acetyl chloride (2.65 ml, 37.2 mmol) at 0°C. The mixture was then stirred at rt for 8 h. The reaction was quenched with saturated NaHC03 and the mixture was extracted with DCM (30 ml) three times. The combined organic phase was washed with brine and dried over anhydrous MgS04, filtered and concentrated to give the title compound J.i (5.0 g, 68.9% yield), which was used directly in next step. LCMS (method B): [MH]+ = 188, tR = 1.64 min. |
| 68.9% |
With pyridine In dichloromethane at 0 - 20℃; for 8h; |
Quinolin-6-yl acetate (A.i)
To a solution of quinolin-6-ol (4.5 g, 31.0 mmol) and pyridine (3.01 ml, 37.2 mmol) in DCM (50 ml) was added acetyl chloride (2.65 ml, 37.2 mmol) at 0°C. The mixture was then stirred at rt for 8 h. The reaction was quenched with saturated NaHC03 and the mixture was extracted with DCM (30 ml) three times. The combined organic phase was washed with brine and dried over anhydrous MgS04, filtered and concentrated to give the title compound (5.0 g, 68.9% yield), which was used directly in next step. LCMS (method B): [M+H]+ = 188, tR = 1.64 min. |
| 68.9% |
With pyridine In dichloromethane at 0 - 20℃; for 8h; |
Quinolin-6-yl acetate (A.i)
To a solution of quinolin-6-ol (4.5 g, 31.0 mmol) and pyridine (3.01 ml, 37.2 mmol) in DCM (50 ml) was added acetyl chloride (2.65 ml, 37.2 mmol) at 0° C. The mixture was then stirred at rt for 8 h. The reaction was quenched with saturated NaHCO3 and the mixture was extracted with DCM (30 ml) three times. The combined organic phase was washed with brine and dried over anhydrous MgSO4, filtered and concentrated to give the title compound (5.0 g, 68.9% yield), which was used directly in next step. LCMS (method B): [M+H]+=188, tR=1.64 min. |
| 68.9% |
With pyridine In dichloromethane at 0 - 20℃; for 8h; |
Quinolin-6-yl acetate (J.i)
To a solution of quinolin-6-ol (4.5 g, 31.0 mmol) and pyridine (3.01 ml, 37.2 mmol) in DCM (50 ml) was added acetyl chloride (2.65 ml, 37.2 mmol) at 0° C. The mixture was then stirred at rt for 8 h. The reaction was quenched with saturated NaHCO3 and the mixture was extracted with DCM (30 ml) three times. The combined organic phase was washed with brine and dried over anhydrous MgSO4, filtered and concentrated to give the title compound J.i (5.0 g, 68.9% yield), which was used directly in next step. LCMS (method B): [MH]+=188, tR=1.64 min. |
| 52% |
With pyridine In dichloromethane at 0 - 20℃; |
188.1 Preparation of quinoline-6-acetate
To 100ml round bottom three-necked flask was added dry dichloromethane 50ml, 4.5 g (31 mmol) of 6-hydroxyquinoline and 2.9 g (37 mmol) of pyridine were sequentially added with stirring, and the mixture was cooled to 0 ° C, Acetyl chloride 2.9g (37mmol) was slowly added dropwise. When the addition was completed, the mixture was stirred at room temperature and allowed to react overnight. After treatment, the reaction solution was poured into 200ml of cold saturated aqueous sodium carbonate, the organic phase was separated, The aqueous phase was extracted with methylene chloride 50ml * 2, the combined organic phase was washed with saturated saline, dried to give the product 3g, Yield 52%, a step directly. |
|
With N-ethyl-N,N-diisopropylamine In tetrahydrofuran at 25℃; |
|

 Chemistry
Chemistry
 Pharmaceutical Intermediates
Pharmaceutical Intermediates
 Inhibitors/Agonists
Inhibitors/Agonists
 Material Science
Material Science














 For Research Only
For Research Only
 110K+ Compounds
110K+ Compounds
 Competitive Price
Competitive Price
 1-2 Day Shipping
1-2 Day Shipping