Alternatived Products of [ 27153-17-9 ]
Product Details of [ 27153-17-9 ]
| CAS No. : | 27153-17-9 |
MDL No. : | MFCD00086773 |
| Formula : |
C8H9NO2
|
Boiling Point : |
- |
| Linear Structure Formula : | - |
InChI Key : | - |
| M.W : |
151.16
|
Pubchem ID : | - |
| Synonyms : |
|
Safety of [ 27153-17-9 ]
| Signal Word: | |
Class: | |
| Precautionary Statements: | |
UN#: | |
| Hazard Statements: | |
Packing Group: | |
Application In Synthesis of [ 27153-17-9 ]
* All experimental methods are cited from the reference, please refer to the original source for details. We do not guarantee the accuracy of the content in the reference.
- Upstream synthesis route of [ 27153-17-9 ]
- Downstream synthetic route of [ 27153-17-9 ]
- 1
-
 [ 153968-39-9 ]
[ 153968-39-9 ]

-
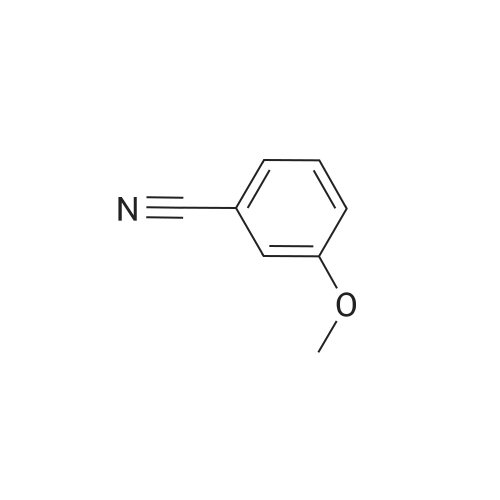 [ 1527-89-5 ]
[ 1527-89-5 ]

-
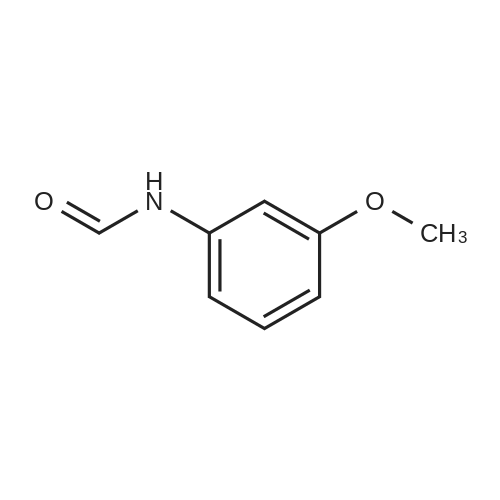 [ 27153-17-9 ]
[ 27153-17-9 ]

-
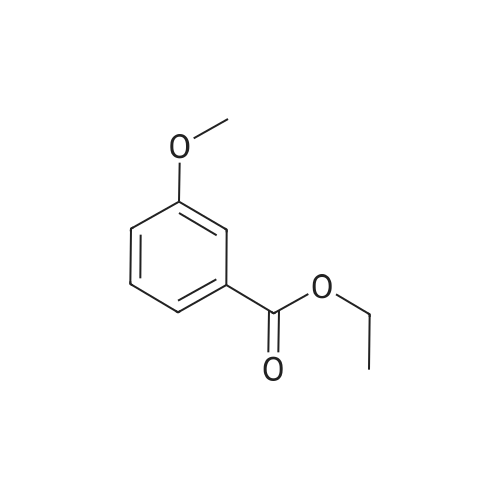 [ 10259-22-0 ]
[ 10259-22-0 ]

-
diethyl m-methoxybenzoylphosphoramidate
[ No CAS ]
- 2
-
 [ 153968-39-9 ]
[ 153968-39-9 ]

-
 [ 1527-89-5 ]
[ 1527-89-5 ]

-
 [ 27153-17-9 ]
[ 27153-17-9 ]

-
 [ 10259-22-0 ]
[ 10259-22-0 ]

-
 [ 536-90-3 ]
[ 536-90-3 ]
- 3
-
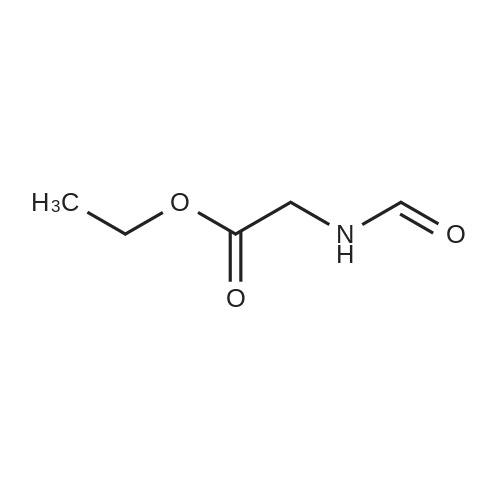 [ 3154-51-6 ]
[ 3154-51-6 ]

-
 [ 27153-17-9 ]
[ 27153-17-9 ]

-
4-ethoxycarbonyl-1-(3-methoxyphenyl)imidazole
[ No CAS ]
| Yield | Reaction Conditions | Operation in experiment |
| 96% |
Stage #1: ethyl N-formylglycinate; 3-Methoxy-formanilid With triethylamine; trichlorophosphate In dichloromethane at 0℃;
Stage #2: In tetrahydrofuran Heating; Further stages.; |
|
- 4
-
 [ 27153-17-9 ]
[ 27153-17-9 ]

-
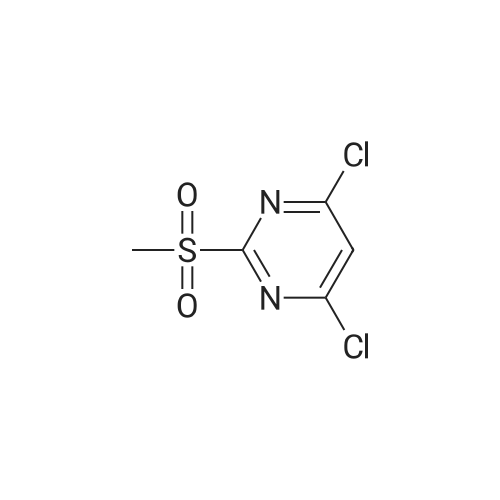 [ 4489-34-3 ]
[ 4489-34-3 ]

-
 [ 1450821-87-0 ]
[ 1450821-87-0 ]
- 5
-
 [ 27153-17-9 ]
[ 27153-17-9 ]

-
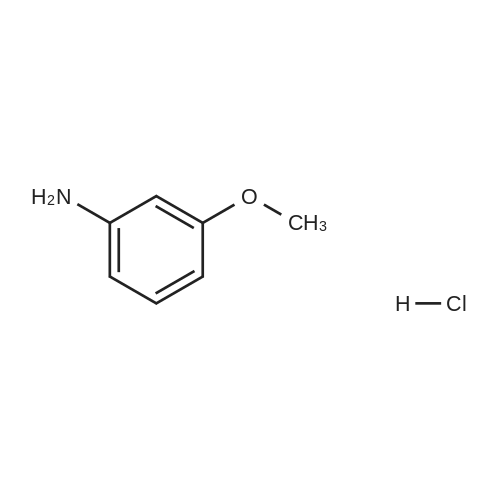 [ 27191-09-9 ]
[ 27191-09-9 ]
- 6
-
 [ 124-38-9 ]
[ 124-38-9 ]

-
 [ 536-90-3 ]
[ 536-90-3 ]

-
 [ 27153-17-9 ]
[ 27153-17-9 ]
| Yield | Reaction Conditions | Operation in experiment |
| 88% |
With dimethylamine borane In N,N-dimethyl-formamide at 60℃; for 12h; Autoclave; |
2.3. Experimental procedure for the synthesis of formamides
General procedure: N-formylation of amines by CO2 and DMAB in the presence of UiO-66 as a catalyst was carried out in autoclave. The synthesis of formamides is as follows, Amines (5.0 mmol), UiO-66 (10 mg), DMAB (1.5 equiv.), DMF (7 mL) were added in the reactor at room temperature, reactor was closed, flushed two to three times by CO2 and 1 MPa CO2 pressure was introduced into reactor, then the reactor was heated to required temperature with continuous stirring. After the reaction was complete, the reactor was cooled, and the CO2 pressure was carefully discharged from the autoclave. The UiO-66 MOF was separated by centrifugation. The resultant mixture was concentrated by rotary evaporator and subjected to the column chromatography with ethyl acetate-petroleum ether used as an eluent to obtain the product. All the products are confirmed by GCMS. |
| 88% |
With ZnO nanoparticles supported on L-serine; poly(methylhydrosiloxane) In 1,4-dioxane at 80℃; for 20h; High pressure; Sealed tube; |
2.1.3. Synthesis of formamides
General procedure: Amines (1.0 mmol), L-Serine(at)ZnO (20 mg), PMHS (2 mmol), DMF(5 mL) were added into high-pressure reactor and sealed the reactor then 15 bar CO2 pressure was maintained in reactor, heated to desiredtemperature, kept for 20 h and cooled the reactor and pressure wasslowly discharged from the high-pressure reactor. The AAs(at)ZnO wasregained by centrifugation dried and reused for next recycled run. Theremaining solvent was concentrated in vacuo and product was separatedby column chromatography and analysed by using the GC as wellas GCMS and NMR analysis. |
| 86% |
With dimethylamine borane In water at 100℃; for 24h; Heating; Green chemistry; |
2.2 Experimental Procedure for the Synthesis of Benzimidazoles
General procedure: Synthesis of benzimidazole from o-phenylenediamine byCO2and DMAB in presence of Cu(at)U-g-C3N4 was carriedout in high pressure reactor equipped with an overheadstirrer. In a general experiment for the synthesis ofbenzimidazole, o-phenylenediamine (1.00mmol), DMAB(3mmol), PC:H2O (3 mL:1.5mL), Cu(at)U-g-C3N4 (20mg)were loaded into the reactor at room temperature, reactorwas sealed, flushed three times with CO2and 2.5MPa CO2pressure was loaded in to reactor, heated to required temperaturewith stirring (600rpm). After completion of thereaction, the reactor was cooled to room temperature andthe pressure was slowly released. The catalyst was separatedby fltration, washed with ethyl acetate and water.The combined mixture was concentrated in vacuo and theproducts were purified by the column chromatographywith silica gel of 100-200 mesh size and petroleum etherethylacetate used as an eluent to aford pure products The spectroscopic data of all the products werematching with those reported in the literature. |
| 84% |
With sodium phenyltrihydroxyborate; phenylsilane In diethylene glycol dimethyl ether at 60℃; for 12h; Sealed tube; Glovebox; Autoclave; |
|
| 58% |
With 1,10-Phenanthroline; phenylsilane; zinc diacetate In acetonitrile at 25℃; for 1h; Autoclave; |
|
| 83 %Chromat. |
With dimethylamine borane In neat (no solvent) at 100℃; for 6h; Autoclave; Green chemistry; |
|
| 90 %Chromat. |
Stage #1: carbon dioxide; m-Anisidine In acetonitrile Schlenk technique;
Stage #2: With phenylsilane In acetonitrile at 50℃; Schlenk technique; |
|
| 76 % |
With [Cp*CoCl2]2; phenylsilane; potassium <i>tert</i>-butylate; C16H20N5(1+)*Br(1-) In dichloromethane; acetonitrile at 20℃; Inert atmosphere; |
|
| 73 % |
With phenylsilane; potassium <i>tert</i>-butylate; zinc diacetate; C16H20N5(1+)*Br(1-) In acetonitrile at 20℃; |
|

Reference:
[1]Phatake, Vishal V.; Mishra, Ashish A.; Bhanage, Bhalchandra M.
[Inorganica Chimica Acta, 2020, vol. 501]
[2]Ahire, Jayendra P.; Bhanage, Bhalchandra M.; Phatake, Vishal V.
[Molecular catalysis, 2020, vol. 492]
[3]Phatake, Vishal V.; Bhanage, Bhalchandra M.
[Catalysis Letters, 2019, vol. 149, # 1, p. 347 - 359]
[4]Du, Chen-Xia; Huang, Zijun; Jiang, Xiaolin; Li, Yuehui; Makha, Mohamed; Wang, Fang; Zhao, Dongmei
[Green Chemistry, 2020, vol. 22, # 16, p. 5317 - 5324]
[5]Zhang, Qiao; Lin, Xiao-Tao; Fukaya, Norihisa; Fujitani, Tadahiro; Sato, Kazuhiko; Choi, Jun-Chul
[Green Chemistry, 2020, vol. 22, # 23, p. 8414 - 8422]
[6]Nale, Deepak B.; Rath, Dharitri; Parida; Gajengi, Aravind; Bhanage, Bhalchandra M.
[Catalysis science and technology, 2016, vol. 6, # 13, p. 4872 - 4881]
[7]Zhu, Kaixun; Li, Yuncong; Li, Zhengyi; Liu, Yixuan; Wu, Hongguo; Li, Hu
[Chemical Communications, 2022, vol. 58, # 91, p. 12712 - 12715]
[8]Siddique, Misba; Boity, Biswaranjan; Rit, Arnab
[Organometallics, 2023, vol. 42, # 12, p. 1395 - 1403]
[9]Sahoo, Sangita; Manna, Subarna; Rit, Arnab
[Catalysis science and technology, 2023, vol. 13, # 11, p. 3344 - 3350]

 Chemistry
Chemistry
 Pharmaceutical Intermediates
Pharmaceutical Intermediates
 Inhibitors/Agonists
Inhibitors/Agonists
 Material Science
Material Science















 For Research Only
For Research Only
 110K+ Compounds
110K+ Compounds
 Competitive Price
Competitive Price
 1-2 Day Shipping
1-2 Day Shipping











