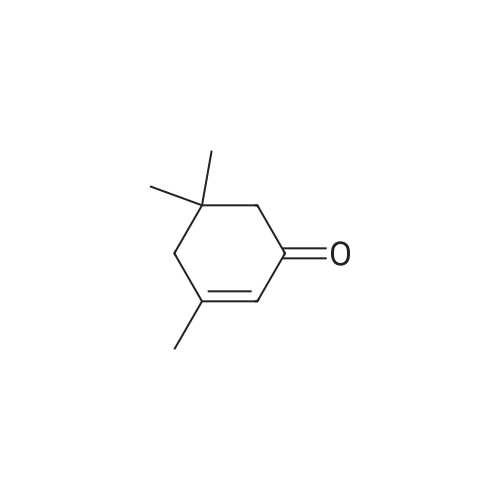
| 66% |
With (2S,5S)-5-benzyl-3-methyl-2-(5-methyl-furan-2-yl)-imidazolidin-4-one; trichloroacetic acid In diethyl ether at 4℃; for 48h; |
|
|
With polymethylhydrosiloxane; copper diacetate; (R)-(-)-3,5-di-tert-butyl-4-methoxy-SEGPHOS In toluene at 20℃; for 1h; |
|
|
With (R)-((4,4’-bi-1,3-benzodioxole)-5,5’-diyl)bis(bis(3,5-di-t-butyl-4-methoxyphenyl))phosphine; polymethylhydrosiloxane; copper diacetate In toluene at 60℃; for 0.166667h; microwave irradiation; |
|
|
With RhH(CO)2[(R,R)-Chiraphos]; hydrogen In tetrahydrofuran at 60℃; for 72h; Inert atmosphere; optical yield given as %ee; |
|
|
With [(4S)-4-[5-bis(3,5-ditert-butyl-4-methoxy-phenyl)phosphanyl-1,3-benzodioxol-4-yl]-4,5,6,7-tetrahydro-1,3-benzodioxol-5-yl]-bis(3,5-ditert-butyl-4-methoxy-phenyl)phosphane; copper diacetate In toluene at 20℃; for 1.5h; |
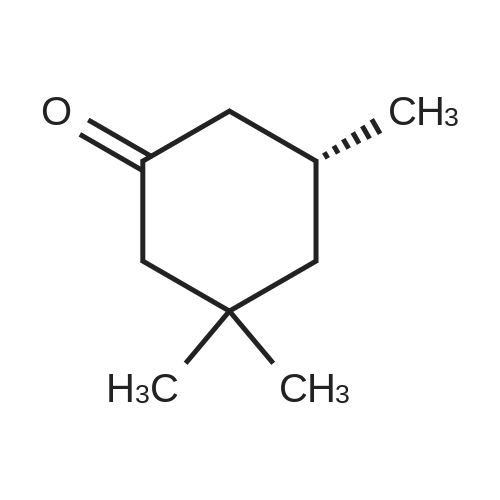
100.1 Step 1: (5R)-3,3,5-Trimethylcyclohexanone
To a round bottom flask purged with nitrogen was added copper (II) acetate (14.53 mg, 0.08000 mmol), [(4S)-4-[5-bis(3,5-ditert-butyl-4-methoxy-phenyl)phosphanyl-1,3-benzodioxol-4-yl]-4,5,6,7-tetrahydro-1,3-benzodioxol-5-yl]-bis(3,5-ditert-butyl-4-methoxy-phenyl)phosphane (47.34 mg, 0.04000 mmol), and toluene (27.64 mL). The reaction was stirred at room temperature for 90 minutes. Polymethylhydrosiloxane (4.8 mL) was added and the reaction was allowed to stir at room temperature for 30 minutes. 3,5,5-Trimethylcyclohex-2-en-1-one (5.528 g, 6.009 mL, 40.00 mmol) was then added dropwise and the reaction was allowed to stir for 90 minutes at room temperature. The reaction was poured into a solution of 3M sodium hydroxide and allowed to stir vigorously for 2 hours. The crude reaction was extracted with ethyl acetate and dried over sodium sulfate. The solvent was removed and the crude product was taken onto the next step without further purification to give (5R)-3,3,5-trimethylcyclohexanone (5.1 g, 36 mmol, 91%) ESI-MS m/z calc. 140.12012. found 141.2 (M+1)+. Retention time: 1.44 minutes was a pale oil. |
| > 99 % ee |
With glucose-6-phosphate In dimethyl sulfoxide at 24℃; for 3h; Sealed tube; Darkness; Enzymatic reaction; |
|
|
With copper diacetate; [(4S)-4-[5-bis(3,5-di-tert-butyl-4-methoxyphenyl)phosphanyl-1,3-benzodioxol-4-yl]-4,5,6,7-tetrahydro-1,3-benzodioxol-5-yl]-bis(3,5-di-tert-butyl-4-methoxyphenyl)phosphane In toluene at 20℃; for 1.5h; |
100.1 Step 1: (5R )-3,3,5-trimethylcyclohexanone
Add copper(II) acetate (14.53mg, 0.08000mmol) to a round bottom flask purged with nitrogen,[(4S )-4-[5-bis(3,5-di-tert-butyl-4-methoxy-phenyl)Phosphanyl-1,3-benzodioxol-4-yl]-4,5,6,7-tetrahydro-1,3-benzodioxol-5- Yl]-bis(3,5-di-tert-butyl-4-methoxy-phenyl)phosphane (47.34 mg, 0.04000 mmol) and toluene (27.64 mL).The reaction was stirred at room temperature for 90 minutes.Polymethylhydrosiloxane (4.8 mL) was added and the reaction was stirred at room temperature for 30 minutes.Then 3,5,5-trimethylcyclohex-2-en-1-one (5.528 g, 6.009 mL, 40.00 mmol) was added dropwise and the reaction was stirred at room temperature for 90 minutes.The reaction was poured into a3M sodium hydroxide solution and stirred vigorously for 2 hours.The crude reaction was extracted with ethyl acetate and dried over sodium sulfate. The solvent was removed and the crude product was used in the next step without further purification to obtain (5R )-3,3,5-trimethylcyclohexanone (5.1 g, 36 mmol, 91%) as a light oil. |

 Chemistry
Chemistry
 Pharmaceutical Intermediates
Pharmaceutical Intermediates
 Inhibitors/Agonists
Inhibitors/Agonists
 Material Science
Material Science













 For Research Only
For Research Only
 110K+ Compounds
110K+ Compounds
 Competitive Price
Competitive Price
 1-2 Day Shipping
1-2 Day Shipping