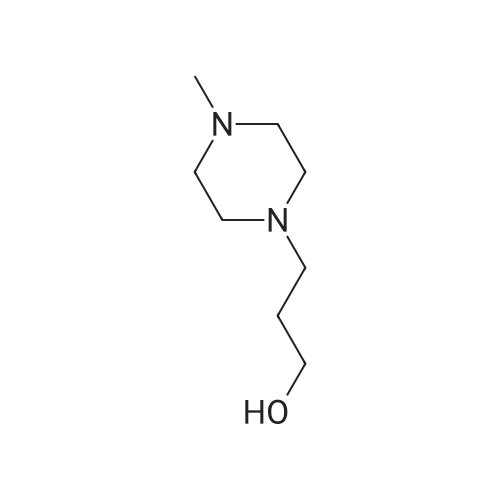
| 90% |
With 2,3,4,5,7,8,9,10-octahydropyrimido[1,2-a]azepin-1-ium acetate at 20℃; for 1.5h; Neat (no solvent); |
|
| 87% |
With ionic liquid [bmim]BF4 stabilized magnetic cobalt nanoparticles In neat (no solvent) at 20℃; for 0.583333h; |
3.2 General procedure for aza-Michael and thia-Michael additions
General procedure: 1mmol of amine or thiol and 1mmol of activated α,β-unsaturated compound (Michael acceptor) were stirred together in the presence of 5mol% (0.1mmol) of stabilized cobalt nanoparticles for an appropriate time (Scheme1). Following the completion of the reaction as indicated by TLC (hexane: ethyl acetate), dichloromethane was added to the reaction mixture and the cobalt catalyst was recovered by applying a magnetic field and decanting rest of the reaction mixture into another flask from where solvent was evaporated off and final product was isolated. Further purification was carried out by column chromatography on silica gel using ethyl acetate/petroleum ether as eluent. Structural assignments of the products were done based on their 1H NMR, 13C NMR and mass spectroscopic analysis. |
|
With methanol |
|
|
at 0 - 50℃; |
A
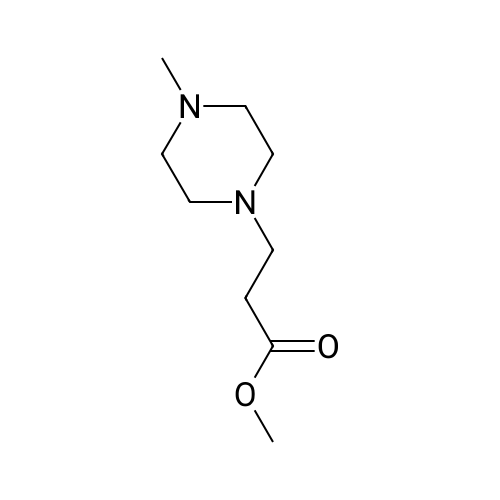
In a 500-mL 3-neck round bottom flask, 150.Og (1.7 moles) of methyl acrylate is charged. The reactor is cooled to O0C using an ice bath. Then, 77.Og (0.77 moles) of 1 -methyl piperazine are added slowly to prevent overheating of the reaction mass. The temperature should be maintained below 5O0C by controlling the addition rate of 1 -methyl piperazine. Overheating may cause polymerization of the excess methyl acrylate present in the reactor. Once the addition is completed, the mixture is agitated using a magnetic stirring bar and the temperature adjusted to 5O0C for at least 16 hours. Then, the reaction is allowed to cool to ambient temperature and the excess methyl acrylate is removed under vacuum. The final product is a light brown liquid at ambient temperature. The transformation is monitored with thin layer chromatography (TLC) using 9:1 CHCl3MeOH. Complete conversion is apparent from the lack of olefinc protons in the 1H-NMR spectra. 1H-NMR (300 MHz, CDCl3): δ 3.20 (s, 3H), 2.12 (t, 7.3Hz, 2H), 1.94 (m, 10H), 1.71 (s, 3H). 13C-NMR (75MHz, CDCl3): δ 171.31, 54.00, 52.41, 51.78, 50.22, 44.92, 30.96. |
| 99.2 %Chromat. |
With crosslinked polymer with a benzyl(triphenyl)phosphonium ionic liquid at 25℃; for 0.0666667h; |
|
|
With Pseudomonas fluorescence lipase immobilized on hyroxypropyl methyl cellulose support In toluene at 50℃; for 3h; Enzymatic reaction; chemoselective reaction; |
2.4. Reaction procedure and analysis
General procedure: Various β-amino ester moieties were synthesized in a 10 mL glass reaction vessel of 1.2 cm i.d. with a glass lid. The desired Michael acceptor was taken into a reaction vessel and diluted by solvent. Afterwards, Michael donor was added into the reaction vessel and reaction was initiated by addition of immobilized HMC:PFL lipase very soon at a specified temperature. The sample of reaction mixture was analyzed by using gas chromatograph (Perkin Elmer Clarus: 400) having flame ionizing detector (FID) and capillary column. The detector and injector temperature were kept 280 and 50 °C, respectively. The oven temperature of GC was kept at 50 °C for 3 min constant and after that raised with 10 °C/min up to the 280 °C. Moreover, reaction products were also verified by GCMS (Gas-Chromatography-Mass Spectroscopy Shimadzu QP-2010) analysis. |
| 99.4 %Chromat. |
With porous hyper-cross-linked polymer containing 2.87 mmol/g of ionic liquid embedded in framework synthesized from 4-vinylpyridine and p-xylylene dichloride In neat (no solvent) at 25℃; for 0.0666667h; |
The Procedure for aza-Michael Addition
General procedure: Requisite amounts of amines (20 mmol), alkenes (24 mmol) and catalyst (20 mg) were mixed and reacted for definite time intervals at ambient temperature. The GC analysis was conducted to monitor the reaction process. On completion, the catalyst was recycled by filtration. The recycled catalyst was washed with ethanol and dried at 80°C overnight. Activity of the recycled system was carefully investigated. |

 Chemistry
Chemistry
 Pharmaceutical Intermediates
Pharmaceutical Intermediates
 Inhibitors/Agonists
Inhibitors/Agonists
 Material Science
Material Science














 For Research Only
For Research Only
 110K+ Compounds
110K+ Compounds
 Competitive Price
Competitive Price
 1-2 Day Shipping
1-2 Day Shipping