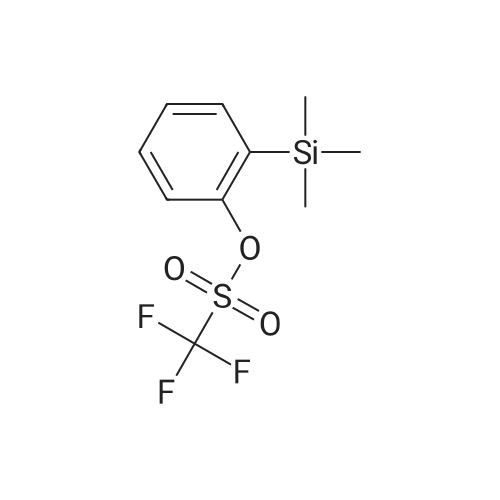
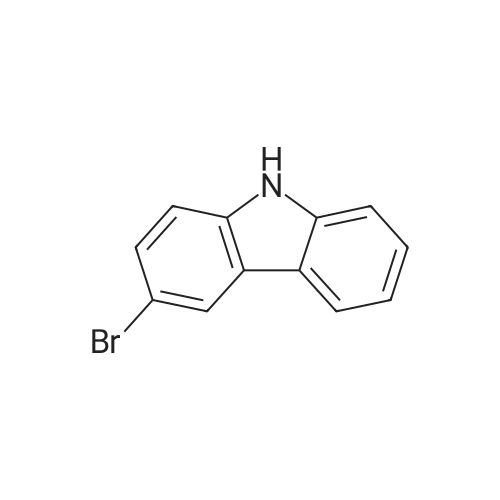
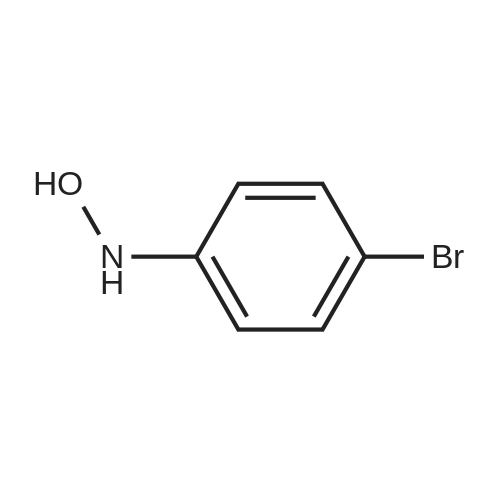
Alternatived Products of [ 3623-23-2 ]
Product Details of [ 3623-23-2 ]
| CAS No. : | 3623-23-2 |
MDL No. : | MFCD02253202 |
| Formula : |
C6H4BrNO
|
Boiling Point : |
- |
| Linear Structure Formula : | - |
InChI Key : | - |
| M.W : |
186.01
|
Pubchem ID : | - |
| Synonyms : |
|
Safety of [ 3623-23-2 ]
| Signal Word: | |
Class: | |
| Precautionary Statements: | |
UN#: | |
| Hazard Statements: | |
Packing Group: | |
Application In Synthesis of [ 3623-23-2 ]
* All experimental methods are cited from the reference, please refer to the original source for details. We do not guarantee the accuracy of the content in the reference.
- Upstream synthesis route of [ 3623-23-2 ]
- Downstream synthetic route of [ 3623-23-2 ]
- 1
-
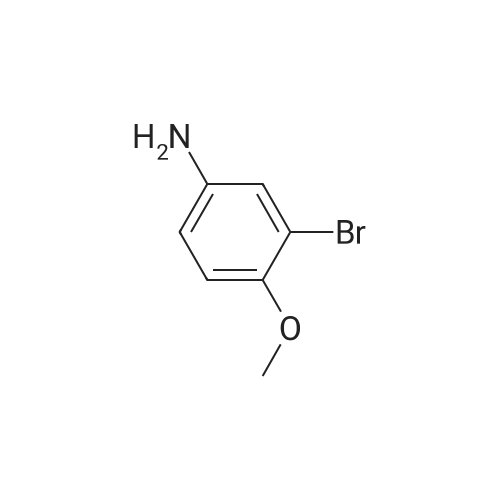 [ 19056-41-8 ]
[ 19056-41-8 ]

-
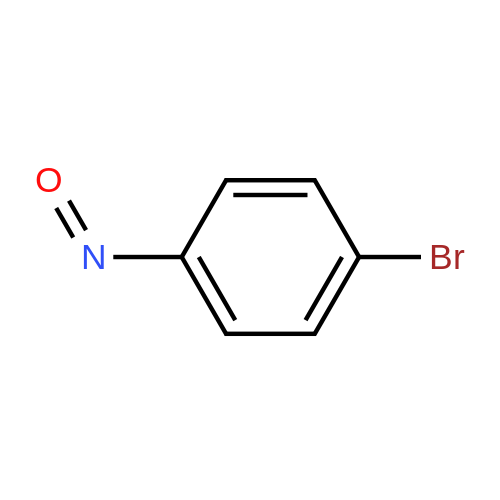 [ 3623-23-2 ]
[ 3623-23-2 ]

-
(3-bromo-4-methoxy-phenyl)-(4-bromo-phenyl)-diazene
[ No CAS ]
- 2
-
 [ 3623-23-2 ]
[ 3623-23-2 ]

-
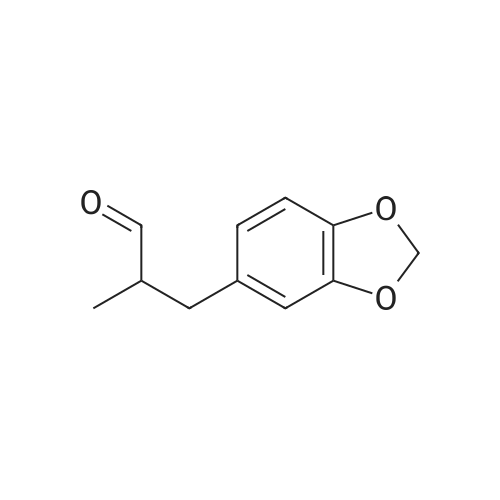 [ 1205-17-0 ]
[ 1205-17-0 ]

-
C17H16BrNO4
[ No CAS ]
- 3
-
 [ 3623-23-2 ]
[ 3623-23-2 ]

-
 [ 1205-17-0 ]
[ 1205-17-0 ]

-
(+)-3-benzo[1,3]dioxol-5-yl-[(4-bromo-phenyl)-hydroxy-amino]-2-methyl-propan-1-ol
[ No CAS ]

-
(-)-3-benzo[1,3]dioxol-5-yl-[(4-bromo-phenyl)-hydroxy-amino]-2-methyl-propan-1-ol
[ No CAS ]
- 4
-
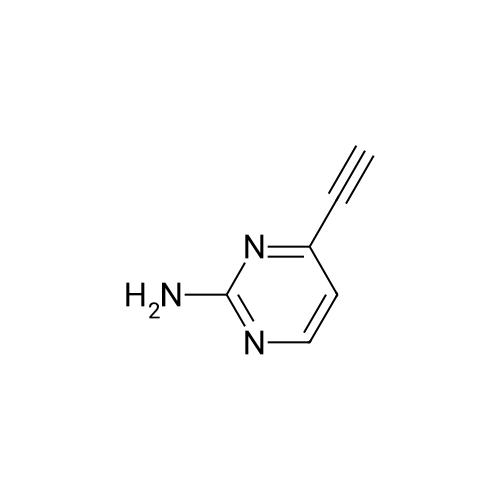 [ 1207175-18-5 ]
[ 1207175-18-5 ]

-
 [ 3623-23-2 ]
[ 3623-23-2 ]

-
Meridianin C
[ No CAS ]
| Yield | Reaction Conditions | Operation in experiment |
| 28% |
In toluene at 80℃; for 7h; Inert atmosphere; |
|
Reference:
[1]Location in patent: experimental part
Tibiletti, Francesco; Simonetti, Marco; Nicholas, Kenneth M.; Palmisano, Giovanni; Parravicini, Matteo; Imbesi, Federico; Tollari, Stefano; Penoni, Andrea
[Tetrahedron, 2010, vol. 66, # 6, p. 1280 - 1288]
- 5
-
 [ 3623-23-2 ]
[ 3623-23-2 ]

-
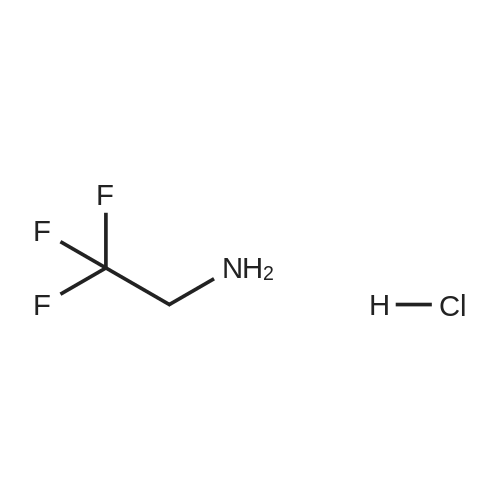 [ 373-88-6 ]
[ 373-88-6 ]

-
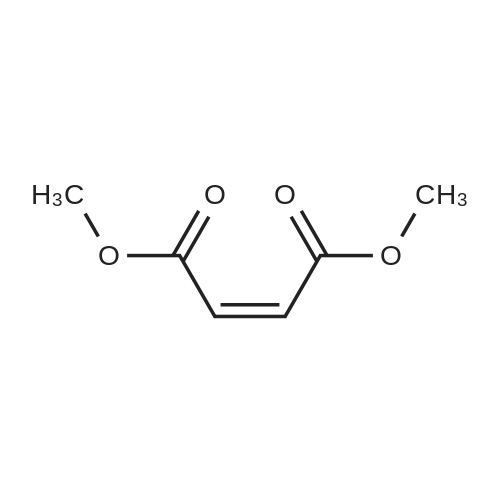 [ 624-48-6 ]
[ 624-48-6 ]

-
dimethyl 2-(4-bromophenyl)-3-(trifluoromethyl)isoxazolidine-4,5-dicarboxylate
[ No CAS ]
- 6
-
 [ 3623-23-2 ]
[ 3623-23-2 ]

-
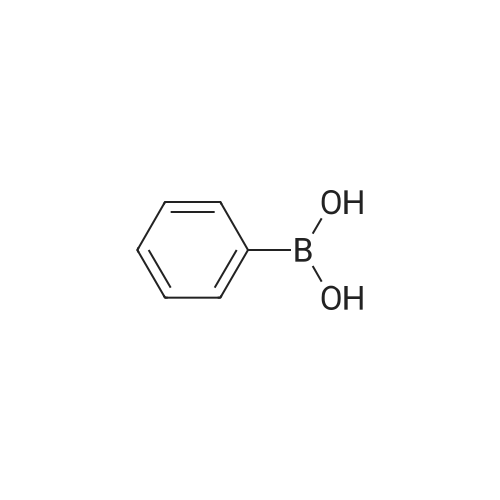 [ 98-80-6 ]
[ 98-80-6 ]

-
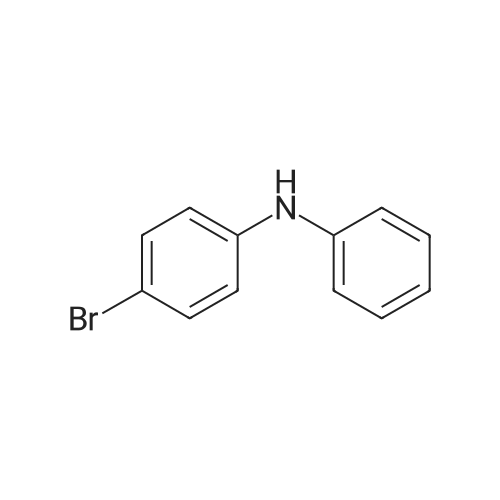 [ 54446-36-5 ]
[ 54446-36-5 ]
| Yield | Reaction Conditions | Operation in experiment |
| 97% |
With triethyl phosphite In toluene at 20℃; for 0.75h; |
|
- 7
-
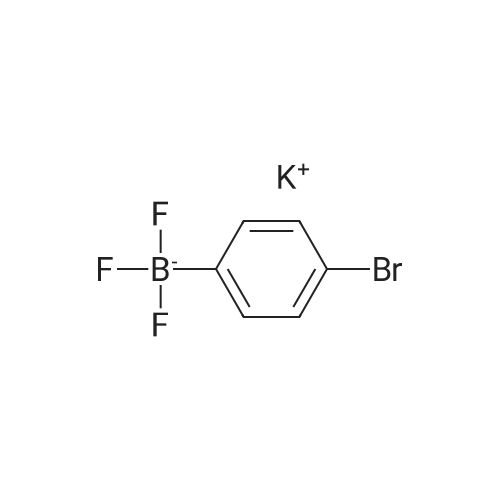 [ 374564-35-9 ]
[ 374564-35-9 ]

-
 [ 3623-23-2 ]
[ 3623-23-2 ]
- 8
-
 [ 1503-99-7 ]
[ 1503-99-7 ]

-
 [ 3623-23-2 ]
[ 3623-23-2 ]

-
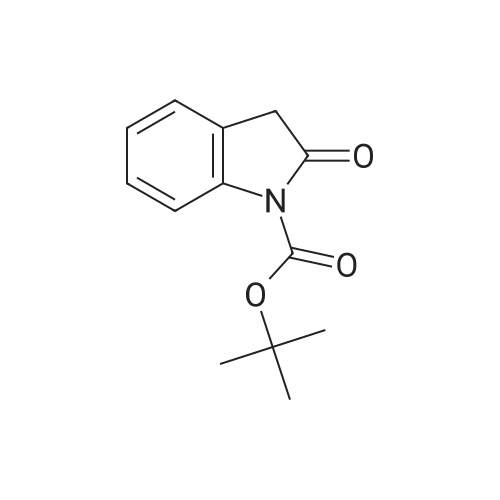 [ 214610-10-3 ]
[ 214610-10-3 ]

-
1'-(tert-butyl)2-ethyl (1S,2R,5R)-2-((4-bromophenyl)amino)-2',3-dioxo-5-phenylspiro[cyclopentane-1,3'-indoline]-1',2-dicarboxylate
[ No CAS ]

-
1'-(tert-butyl)2-ethyl 2-((4-bromophenyl)amino)-2',3-dioxo-5-phenylspiro[cyclopentane-1,3'-indoline]-1',2-dicarboxylate
[ No CAS ]
| Yield | Reaction Conditions | Operation in experiment |
| 61% |
Stage #1: ethyl 3-oxo-5-phenylpent-4-enoate; 4-bromonitrosobenzene With 3-((3,5-bis(trifluoromethyl)benzyl)amino)-4-(((1S)-(6-methoxyquinolin-4-yl)((2S,4S,5R)-5-vinylquinuclidin-2-yl)methyl)amino)cyclobut-3-ene-1,2-dione In dichloromethane at 25℃; for 2h; Molecular sieve;
Stage #2: N-boc-2-oxyindole In dichloromethane at 0℃; for 10h; Molecular sieve; stereoselective reaction; |
|
Reference:
[1]Wang, Chuan-Chuan; Huang, Jian; Li, Xin-Hao; Kramer, Søren; Lin, Guo-Qiang; Sun, Xing-Wen
[Organic Letters, 2018, vol. 20, # 10, p. 2888 - 2891]
- 9
-
 [ 3623-23-2 ]
[ 3623-23-2 ]

-
 [ 373-88-6 ]
[ 373-88-6 ]

-
 [ 106-96-7 ]
[ 106-96-7 ]

-
2-bromo-1-(1-(4-bromophenyl)-3-(trifluoromethyl)aziridin-2-yl)ethan-1-one
[ No CAS ]

-
2-bromo-1-(1-(4-bromophenyl)-3-(trifluoromethyl)aziridin-2-yl)ethan-1-one
[ No CAS ]
- 10
-
 [ 3623-23-2 ]
[ 3623-23-2 ]

-
 [ 373-88-6 ]
[ 373-88-6 ]

-
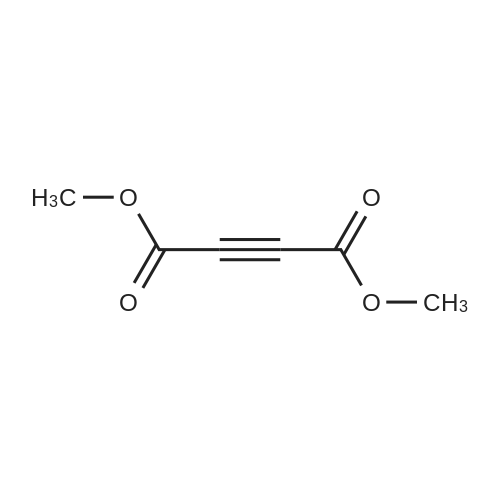 [ 762-42-5 ]
[ 762-42-5 ]

-
methyl 1-(4-bromophenyl)-2-(2-methoxy-2-oxoacetyl)-3-(trifluoromethyl)aziridine-2-carboxylate
[ No CAS ]
- 11
-
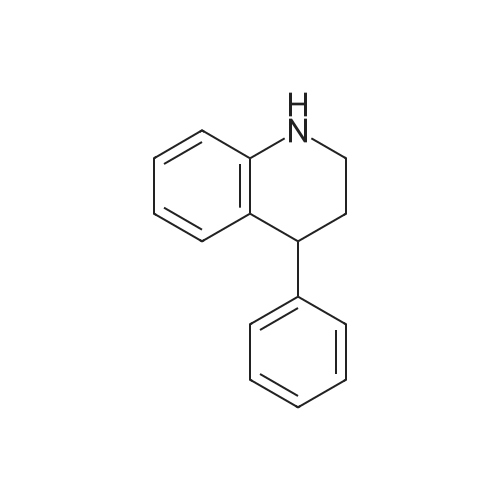 [ 30290-78-9 ]
[ 30290-78-9 ]

-
 [ 3623-23-2 ]
[ 3623-23-2 ]

-
2-((5-bromo-1H-indol-3-yl)(phenyl)methyl)aniline
[ No CAS ]
| Yield | Reaction Conditions | Operation in experiment |
| 50% |
With 4-nitro-benzoic acid In toluene for 18h; Inert atmosphere; |
|
- 12
-
 [ 3623-23-2 ]
[ 3623-23-2 ]

-
 [ 106-49-0 ]
[ 106-49-0 ]

-
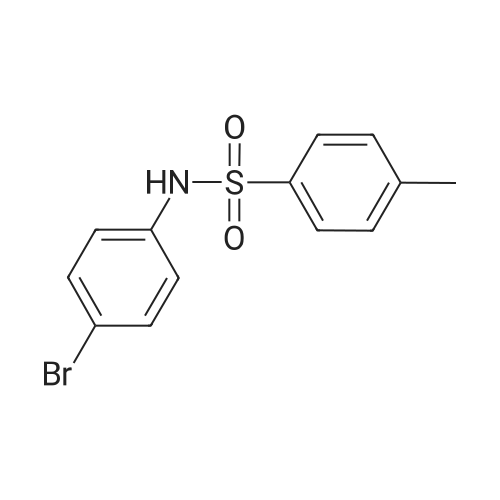 [ 32857-48-0 ]
[ 32857-48-0 ]
| Yield | Reaction Conditions | Operation in experiment |
|
Stage #1: <i>p</i>-toluidine With hydrogenchloride; sodium nitrite In water at 0℃;
Stage #2: With sodium tetrafluoroborate In water
Stage #3: 4-bromonitrosobenzene With cyclohexa-1,4-diene; 1,4-diazabicyclo [2.2.2] octane-1,4-diium-1,4-disulfinate In acetonitrile at 60℃; for 16h; Sealed tube; Inert atmosphere; |
|
Reference:
[1]Wang, Xuefeng; Lin, Yanmei; Liu, Jin-Biao; He, Fu-Sheng; Kuang, Yunyan; Wu, Jie
[Chinese Journal of Chemistry, 2020, vol. 38, # 10, p. 1098 - 1102]
- 13
-
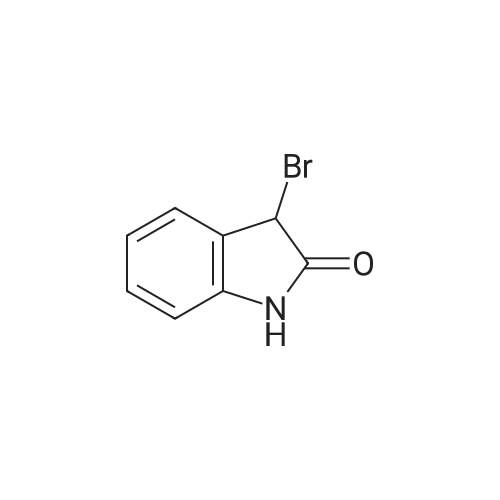 [ 22942-87-6 ]
[ 22942-87-6 ]

-
 [ 3623-23-2 ]
[ 3623-23-2 ]

-
(Z)-4-bromo-N-(2-oxoindolin-3-ylidene)aniline oxide
[ No CAS ]

-
(E)-N-(4-bromophenyl)-2-oxoindolin-3-imine oxide
[ No CAS ]
| Yield | Reaction Conditions | Operation in experiment |
| 86 % de |
With dmap; lithium carbonate In N,N-dimethyl acetamide at 20℃; for 24h; Inert atmosphere; Overall yield = 99percent; Overall yield = 62.8 mg; |
|

 Chemistry
Chemistry
 Pharmaceutical Intermediates
Pharmaceutical Intermediates
 Inhibitors/Agonists
Inhibitors/Agonists
 Material Science
Material Science














 For Research Only
For Research Only
 110K+ Compounds
110K+ Compounds
 Competitive Price
Competitive Price
 1-2 Day Shipping
1-2 Day Shipping