| 83% |
Stage #1: maleiimide; N-benzyl-N-(methoxymethyl)-N-[(trimethylsilyl)methyl]amine With trifluoroacetic acid In dichloromethane at 0 - 20℃;
Stage #2: With sodium hydrogencarbonate In dichloromethane; water
Stage #3: With hydroxylamine In methanol; water at 20℃; for 20h; |
1.1A
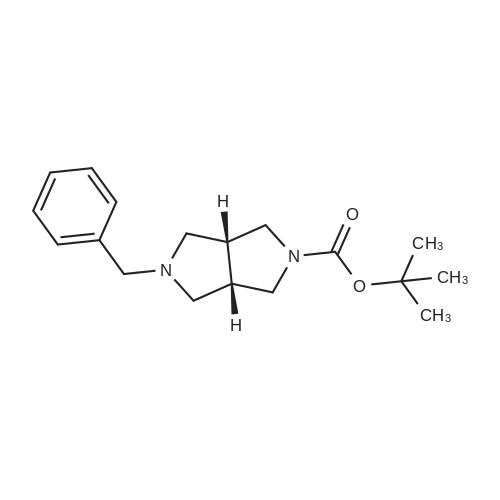
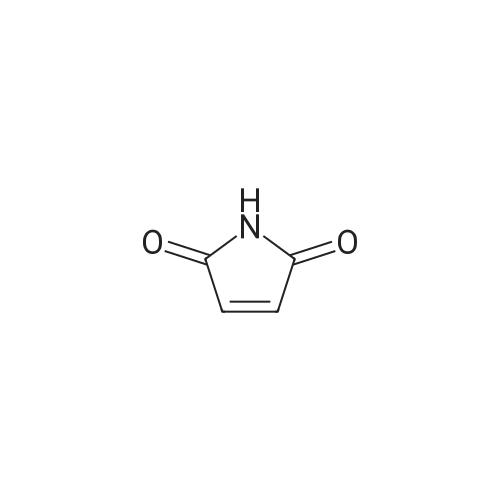
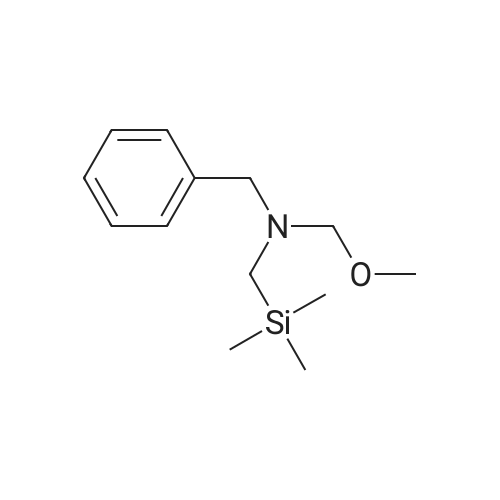
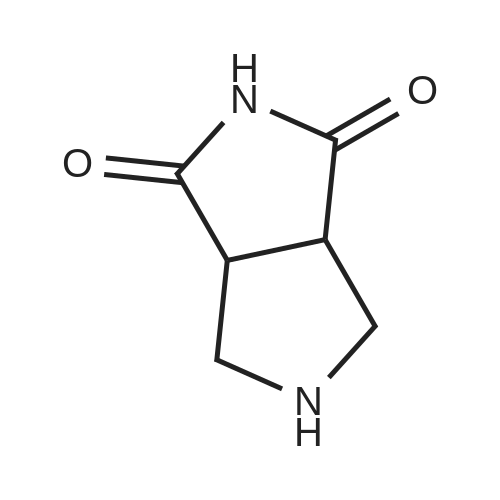
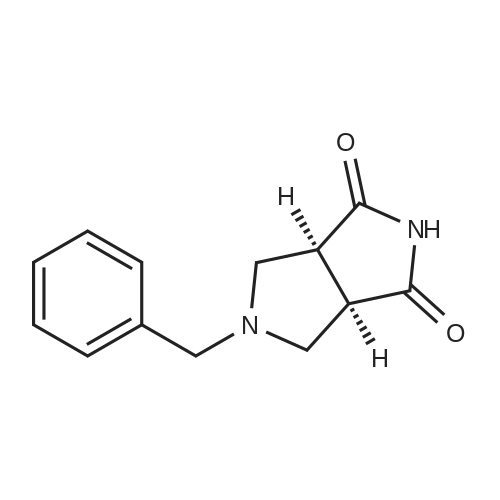
Example 1; N-(3,5-dimethylphenyl)-6-((3aR,6aS)-hexahydropyrrolo[3,4-c]pyrrol-2(1H)-yl)pyrazine-2-carboxamide; Example 1A; (3aR,6aS)-5-benzyltetrahydropyrrolo[3,4-c]pyrrole-1,3(2H,3aH)-dione; A solution of N-benzyl-N-methoxymethyl-N-(trimethylsilylmethyl)amine (76.68 g, 0.29 mol, prepared as in Organic Syntheses (1989), 67, 133-140) in dichloromethane (130 mL) was added to an ice-cooled mixture of maleimide (25.53 g, 0.26 mol) and trifluoroacetic acid (2.2 mL, 0.028 mol) in dichloromethane (350 ml) over 40 min so that the reaction temperature remained between 0-5° C. The resulting bright yellow solution was allowed to warm gradually and stirred at room temperature for 27 h. The mixture was washed with saturated NaHCO3(aq) (80 mL) and the organic phase was dried (MgSO4) and concentrated under vacuum. The residual oil was stirred with 10% EtOAc-heptane (300 mL) for 15 h, and the resulting precipitate was isolated by filtration, washed with 10% EtOAc-heptane (150 mL) and dried under vacuum at 50° C. to provide the crude, title compound as a white solid (54.25 g). This was stirred with methanol (500 mL) and 50% aqueous H2NOH (4.2 mL) at room temperature for 20 h. The mixture was concentrated under vacuum and the residue was taken up in EtOAc (500 mL) and filtered to remove some insoluble material. The filtrate was concentrated under vacuum to leave the pure, title compound as an off-white solid (49.9 g, 83% yield). 1H NMR (300 MHz, CD3OD) δ ppm 2.32-2.43 (m, 2H), 3.18 (d, J=9.8 Hz, 2H), 3.20-3.26 (m, 2H), 3.59 (s, 2H), 7.13-7.35 (m, 5H); MS (DCI/NH3) m/z 231 (M+H)+. |
| 39% |
With trifluoroacetic acid In dichloromethane at 20℃; for 35h; |
5.1
To a solution of 1H-pyrrole-2,5-dione (12.6 g, 130 mmol) in DCM (150 mL) at 0°C was added TFA (1 .1 mL) and a solution of N-benzyl- 1 -methoxy-N-((trimethylsilyl)methyl)5 methanamine (33.9 g, 143 mmol) in DCM (50 mL). The reaction mixture was then stirred atambient temperature for 35 h. The organic layer was washed with sat. NaHCO3 and brine. The organic layer was dried over anhydrous sodium sulfate, filtered and evaporated. The residue was stirred at ethyl acetate / heptane (10 %, 150 mL) overnight. The solid was collected and MeOH1/ NH2OH (aq. 50 %) (2.1 mL) was added. The reaction mixture was stirred at ambient temperatureovernight and then concentrated and the residue was dissolved in ethyl acetate, filtered to remove some insoluble materials. The filtrated was concentrated to provide (3aR,6a5)-5- benzyltetrahydropyrrolo[3,4-c]pyrrole-1,3(2H,3aH)-dione as a light yellow solid (13 g, 39 %). |
| 39% |
With trifluoroacetic acid In dichloromethane at 0 - 20℃; for 35h; |
11.1 Step 1
To a solution of lH-pyrrole-2,5-dione (12.6 g, 130 mmol) in dichloromethane (150 mL) was added TFA (1.1 mL) and a solution of N-benzyl-l-methoxy-N-((trimethylsilyl)methyl)- methanamine (33.9 g, 143 mmol) in dichloromethane (50 mL) at 0 oC dropwise. The mixture was stirred at ambient temperature for 35 h. The organic layer was washed with sat. sodium bicarbonate aqueous solution, washed with brine. The organic layer was dried over anhydrous sodium sulfate, filtered, and evaporated under vacuum. The residue was stirred at ethyl acetate / heptane (1 / 9, 150 mL) overnight. The solid was collected and Methanol/ H20H (aq. 50 %) (2.1 mL) was added. The mixture was stirred at ambient temperature overnight.The solvent was removed and the residue was diluted with ethyl acetate and filtered to remove some insoluble materials. The filtrated was concentrated to give the (3aR,6aS)-5-benzyltetrahydropyrrolo[3,4- c]pyrrole-l,3(2H,3aH)-dione as a light yellow solid (13 g, 39 %). |

 Chemistry
Chemistry
 Pharmaceutical Intermediates
Pharmaceutical Intermediates
 Inhibitors/Agonists
Inhibitors/Agonists
 Material Science
Material Science














 For Research Only
For Research Only
 110K+ Compounds
110K+ Compounds
 Competitive Price
Competitive Price
 1-2 Day Shipping
1-2 Day Shipping