Alternatived Products of [ 639458-46-1 ]
Product Details of [ 639458-46-1 ]
| CAS No. : | 639458-46-1 |
MDL No. : | |
| Formula : |
C12H21NO3
|
Boiling Point : |
- |
| Linear Structure Formula : | - |
InChI Key : | - |
| M.W : |
227.30
|
Pubchem ID : | - |
| Synonyms : |
|
Safety of [ 639458-46-1 ]
| Signal Word: | |
Class: | |
| Precautionary Statements: | |
UN#: | |
| Hazard Statements: | |
Packing Group: | |
Application In Synthesis of [ 639458-46-1 ]
* All experimental methods are cited from the reference, please refer to the original source for details. We do not guarantee the accuracy of the content in the reference.
- Upstream synthesis route of [ 639458-46-1 ]
- Downstream synthetic route of [ 639458-46-1 ]
- 1
-
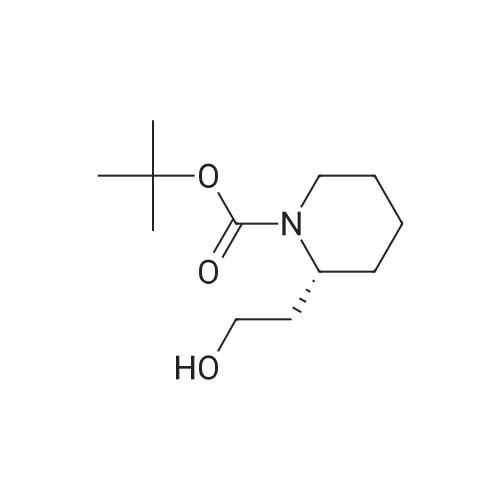
 [ 639458-46-1 ]
[ 639458-46-1 ]

-
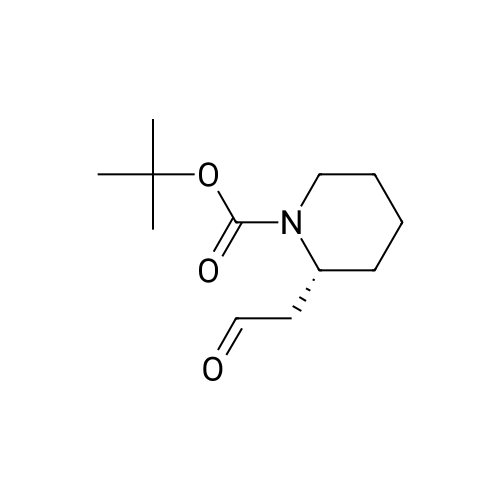
 [ 351410-32-7 ]
[ 351410-32-7 ]
| Yield | Reaction Conditions | Operation in experiment |
|
With sodium chlorite; sodium dihydrogenphosphate; dihydrogen peroxide; In methanol; water; acetonitrile; at 0 - 20℃; for 3.0h;Inert atmosphere; |
To a solution of aldehyde 5 (234 mg, 1.03 mmol) in a mixture of methanol (1 mL), acetonitrile (1 mL), and water (1 mL), KH2PO4 (387 mg, 2.84 mmol), NaClO2 (1.95 mL, 2.15 mmol), and H2O2 30% sol (0.23 mL, 2.05 mmol) were added at 0 C. The reaction was allowed to reach room temperature and stirred for 2 h. Then, 1 M HCl solution was added until pH 3 and a saturated solution of Na2SO3 was added (2 mL). The quenched reaction mixture was extracted with dichloromethane (3×10 mL) and the combined organic layers were dried over anhydrous Na2SO4. Solvents were removed under reduced pressure and the crude mixture was dissolved in toluene (5 mL) and methanol (15 mL). Then, a solution of trimethylsilyldiazomethane (2 M in diethyl ether, 1.1 mL, 2.06 mmol) was added dropwise at 0 C and the reaction was allowed to reach room temperature and stirred for 3 h. Acetic acid (0.5 mL) was added to the reaction mixture and then it was concentrated to dryness. The crude mixture was diluted with diethyl ether (15 mL) and H2O (15 mL), extracted with diethyl ether (3×15 mL) and the combined organic layers were dried over anhydrous Na2SO4. Solvents were removed under reduced pressure and the crude mixture was purified by flash chromatography on silica gel using n-hexane/ethyl acetate (5:1) as eluent to afford 165 mg (62% yield) of a colorless oil |
- 2
-
 [ 87691-87-0 ]
[ 87691-87-0 ]

-
 [ 639458-46-1 ]
[ 639458-46-1 ]

-
(R)-tert-butyl 2-(2-(4-(benzo[d]isothiazol-3-yl)piperazin-1-yl)ethyl)piperidine-1-carboxylate
[ No CAS ]
| Yield | Reaction Conditions | Operation in experiment |
| 89% |
With sodium tris(acetoxy)borohydride; acetic acid; In tetrahydrofuran; at 20℃; for 4h; |
General procedure: To a solution of intermediate 11 (1g, 4.7mmol), 1-(benzothiophen-4-yl)piperazine (1.5g, 7mmol) in THF (30mL) was added sodium triaceoxyborohydride (1.98g, 9.4mmol) and anhydrous AcOH (1mL). After stirring for 4h at room temperature, saturated NaHCO3 was added to the mixture, and the aqueous layer was extracted with AcOEt (3×20mL). The organic layer was washed with water and brine, dried over anhydrous Na2SO4, and concentrated under reduced pressure. The residue obtained was purified by column chromatography on silica gel, using a mixture of CH2Cl2/MeOH (9/0.5 v/v), to give the Boc-protected derivative 24 (1.70g, 4.14mmol) as light oil (yield 88%). The same procedure was used for the synthesis of intermediates 16-37. |

 Chemistry
Chemistry
 Pharmaceutical Intermediates
Pharmaceutical Intermediates
 Inhibitors/Agonists
Inhibitors/Agonists
 Material Science
Material Science















 For Research Only
For Research Only
 110K+ Compounds
110K+ Compounds
 Competitive Price
Competitive Price
 1-2 Day Shipping
1-2 Day Shipping