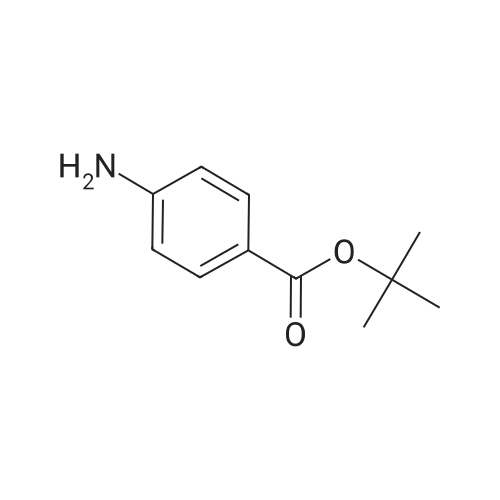
Alternatived Products of [ 769121-30-4 ]
Product Details of [ 769121-30-4 ]
| CAS No. : | 769121-30-4 |
MDL No. : | N/A |
| Formula : |
C13H18N2O3
|
Boiling Point : |
- |
| Linear Structure Formula : | - |
InChI Key : | - |
| M.W : |
250.29
|
Pubchem ID : | - |
| Synonyms : |
|
Safety of [ 769121-30-4 ]
| Signal Word: | |
Class: | N/A |
| Precautionary Statements: | |
UN#: | N/A |
| Hazard Statements: | |
Packing Group: | N/A |
Application In Synthesis of [ 769121-30-4 ]
* All experimental methods are cited from the reference, please refer to the original source for details. We do not guarantee the accuracy of the content in the reference.
- Upstream synthesis route of [ 769121-30-4 ]
- Downstream synthetic route of [ 769121-30-4 ]
- 1
-
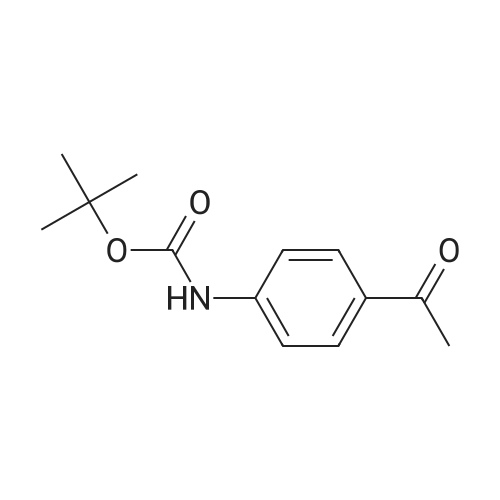 [ 232597-42-1 ]
[ 232597-42-1 ]

-
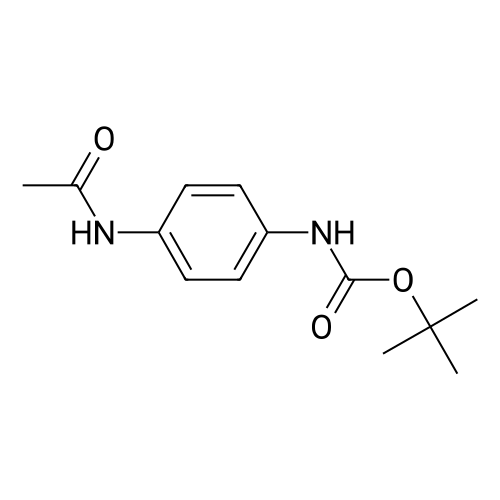 [ 769121-30-4 ]
[ 769121-30-4 ]
| Yield | Reaction Conditions | Operation in experiment |
| 89% |
With zinc(II) chloride; hydroxylamine-O-sulfonic acid In water at 20℃; for 6h; |
Secondary Amides from Ketones; General Procedure
General procedure: To a stirring solution of ZnCl2 (0.05 mmol, 10 mol%) in H2O (2 mL) at r.t. in an open round-bottom flask, ketone 1 (0.5 mmol, 1.0 equiv) wasadded, followed by HOSA (1.5 equiv). The reaction mixture was stirred at the indicated temperature and for the duration indicated in Scheme 2. After completion, the reaction mixture was diluted with EtOAc (15 mL) and washed with sat. aq Na2CO3 (3 × 5 mL). The organic layer was washed with brine (5 mL) and dried over anhydrous Na2SO4.The crude product obtained after removal of all volatiles in vacuo was washed with n-hexane to remove some minor nonpolar impurities. |
| 85% |
With mesitylenesulfonylhydroxylamine In acetonitrile at 20℃; for 6h; |
General procedure for the preparation of amides from ketones
General procedure: To a round bottom flask, equipped with a magnetic stirring bar, was added ketone 1 (0.5 mmol, 1.0 equiv.) and acetonitrile (2 mL) at room temperature. To this stirred solution, freshly prepared O-(Mesitylsulfonyl)hydroxylamine 2 (2.0 equiv.) was added. The reaction mixture was stirred for the specified duration and temperature. The progress of the reaction was monitored by TLC. After completion, the reaction mixture was diluted with ethyl acetate (10 mL) and washed with a saturated aqueous NaHCO3 solution (3 x 5 mL). The combined organic layer was washed with brine solution and dried over anhydrous Na2SO4. Solvent was removed under reduced pressure to get the crude product. The reaction that required elevated temperature was stirred first at room temperature for 2 hours after addition of MSH and then heated at 70 °C for the specified time. |
|
Multi-step reaction with 2 steps
1: sodium acetate; hydroxylamine hydrochloride / water; ethanol / Reflux
2: 2-methoxycarbonylphenylboronic acid; perfluoropinacol; 1,1,1,3',3',3'-hexafluoro-propanol / nitromethane / 24 h / 20 °C |
|
Reference:
[1]Verma, Saumya; Kumar, Puneet; Khatana, Anil K.; Chandra, Dinesh; Yadav, Ajay K.; Tiwari, Bhoopendra; Jat, Jawahar L.
[Synthesis, 2020, vol. 52, # 21, p. 3272 - 3276]
[2]Chandra, Dinesh; Verma, Saumya; Pandey, Chandra Bhan; Yadav, Ajay K.; Kumar, Puneet; Tiwari, Bhoopendra; Jat, Jawahar L.
[Tetrahedron Letters, 2020, vol. 61, # 18]
[3]Mo, Xiaobin; Morgan, Timothy D. R.; Ang, Hwee Ting; Hall, Dennis G.
[Journal of the American Chemical Society, 2018, vol. 140, # 15, p. 5264 - 5271]
Categories

 Chemistry
Chemistry
 Pharmaceutical Intermediates
Pharmaceutical Intermediates
 Inhibitors/Agonists
Inhibitors/Agonists
 Material Science
Material Science














 For Research Only
For Research Only
 110K+ Compounds
110K+ Compounds
 Competitive Price
Competitive Price
 1-2 Day Shipping
1-2 Day Shipping