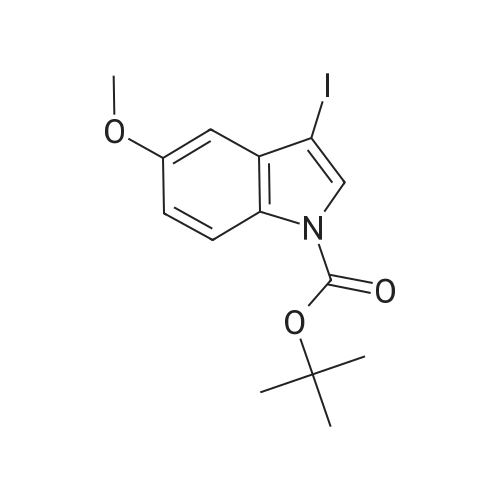
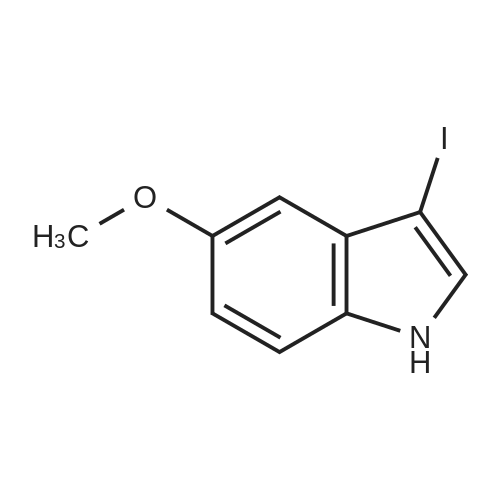
| 92% |
With [bis(acetoxy)iodo]benzene; sodium iodide In water; acetonitrile at 20℃; regioselective reaction; |
|
| 87% |
With potassium hydroxide; iodine In N,N-dimethyl-formamide at 20℃; for 0.45h; |
|
| 84% |
With potassium iodide In acetonitrile at 20℃; for 6h; Electrochemical reaction; Inert atmosphere; regioselective reaction; |
|
| 80% |
With N-iodo-succinimide In dimethyl sulfoxide at 30℃; for 3h; |
General procedure for the synthesis of 3-iodoindoles
General procedure: To a 5 mL round-bottom flask containing 0.1 g of indole dissolved in 2.0 mL of DMSO, NIS (1.05 equiv) was added, and the reaction mixture was stirred for 0.5-24 h (cf. Table 3). After completion of the reaction as monitored by TLC, the reaction mixture was diluted with 20 mL of ethyl acetate, and it was washed with saturated solution of Na2S2O3. The organic phase was subsequently dried over anhyd. sodium sulfate and the solvent was removed in vacuo. The resultant crude product compound was subjected silica gel column chromatography to isolate the product; in the case of 5-methoxy-3-iodoindole, neutral alumina was employed. |
|
With pyridine; Iodine monochloride In dichloromethane at 0 - 20℃; for 0.75h; |
|
|
With iodine; potassium hydroxide In N,N-dimethyl-formamide at 20℃; |
|
|
With iodine; potassium hydroxide In N,N-dimethyl-formamide at 20℃; for 0.75h; |
|
|
With iodine; potassium hydroxide In N,N-dimethyl-formamide |
|
|
Stage #1: 5-methoxylindole With potassium hydroxide In N,N-dimethyl-formamide at 20℃; for 0.333333h;
Stage #2: With ammonium hydroxide; potassium pyrosulfite; iodine In N,N-dimethyl-formamide for 1h; Cooling with ice; |
|
|
Stage #1: 5-methoxylindole With potassium hydroxide In N,N-dimethyl-formamide at 20℃; for 0.333333h;
Stage #2: With iodine In N,N-dimethyl-formamide at 20℃; for 0.75h; |
2 2.2 General procedure for iodination of indole
General procedure: To a solution of indole or its derivatives (1.25mmol) in DMF (6mL) was added KOH (2 equiv) and allowed to stir at room temperature for 20 min. I2 (1 equiv) dissolved in DMF (1 mL) was added and the solution was stirred for an additional 45 min. The reaction mixture was poured into ice water (40 mL) and the precipitate was collected by filtration, washed with water and dried by azeotropic distillation with toluene to yield the 3-iodoindole derivatives. Because of their instability, these derivatives were carried forward to the next step. |
|
With iodine; potassium hydroxide In N,N-dimethyl-formamide at 15℃; for 2h; |
[1208] Preparation of DD-2. To a solution of 5-methoxy-1H-indole (5 g, 33.97 mmol, 1 eq) in DMF were added KOH (4.77 g, 84.93 mmol, 2.5 eq) and I2 (8.62 g, 33.97 mmol, 6.84 mL, 1 eq) in DMF (40 mL) at 15°C. The mixture was stirred at 15°C. for 2 h to give a brown solution. TLC (PE:EA=4:1) showed the reaction was completed. The reaction was poured into ice and water (400 mL) containing ammonia (0.5%) and sodium sulfite (0.1% aqueous solution), and then extracted with MTBE (50 mL4). The combined organic layers were dried over Na2SO4, filtered and concentrated to give DD-2 . 1H NMR (400 MHz, CDCl3) δ=8.54 (br s, 1H), 7.20-7.12 (m, 2H), 6.85-6.75 (m, 2H), 3.81 (s, 3H). |

 Chemistry
Chemistry
 Pharmaceutical Intermediates
Pharmaceutical Intermediates
 Inhibitors/Agonists
Inhibitors/Agonists
 Material Science
Material Science













 For Research Only
For Research Only
 110K+ Compounds
110K+ Compounds
 Competitive Price
Competitive Price
 1-2 Day Shipping
1-2 Day Shipping