Alternatived Products of [ 892663-09-1 ]
Product Details of [ 892663-09-1 ]
| CAS No. : | 892663-09-1 |
MDL No. : | |
| Formula : |
C22H18Cl2FNO3
|
Boiling Point : |
- |
| Linear Structure Formula : | - |
InChI Key : | - |
| M.W : |
434.29
|
Pubchem ID : | - |
| Synonyms : |
|
Safety of [ 892663-09-1 ]
| Signal Word: | |
Class: | |
| Precautionary Statements: | |
UN#: | |
| Hazard Statements: | |
Packing Group: | |
Application In Synthesis of [ 892663-09-1 ]
* All experimental methods are cited from the reference, please refer to the original source for details. We do not guarantee the accuracy of the content in the reference.
- Downstream synthetic route of [ 892663-09-1 ]
- 1
-
 [ 892663-06-8 ]
[ 892663-06-8 ]

-
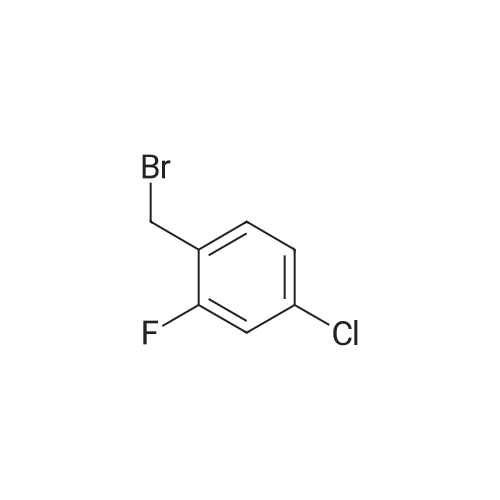 [ 71916-82-0 ]
[ 71916-82-0 ]

-
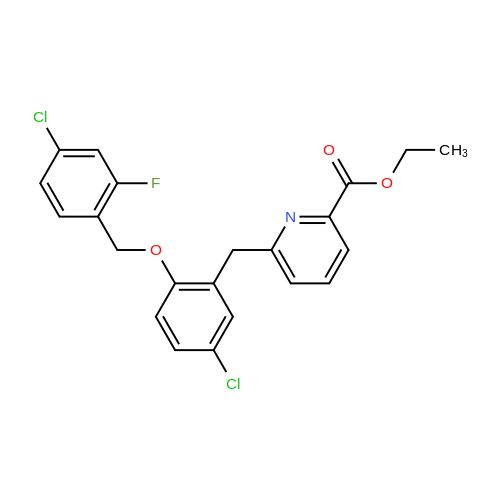 [ 892663-09-1 ]
[ 892663-09-1 ]
| Yield | Reaction Conditions | Operation in experiment |
|
With potassium carbonate In acetone at 50℃; for 3 - 4h; |
1
General procedure 1; EPO Ethyl 6-f(5-chloro-2-(r(4-chloro-2-fluorophenyl)methylloxy)phenvnmethyll-2- pyridinecarboxylate; A mixture of ethyl θ-^S-chloro^-hydroxyphenyOmethylJ^-pyridinecarboxylate (92mg, 0.31 mmol), potassium carbonate (109mg, 2.5 equivalents) and 4-chloro-2-fluorobenzyl bromide (78mg, 1.1 equivalents) in acetone (~6ml) was heated at 500C for 3-4 hours until there was no more SM (starting material). Cooled down, filtered off and washed with acetone. LC/MS okay, Rt 4.05. Purified by SPE, product came off with -20% ethyl acetate in iso-hexane. 108mg of a white solid. Rt = 4.05 [MH+] 434, 437. |
- 2
-
 [ 892663-03-5 ]
[ 892663-03-5 ]

-
 [ 71916-82-0 ]
[ 71916-82-0 ]

-
 [ 892663-09-1 ]
[ 892663-09-1 ]
| Yield | Reaction Conditions | Operation in experiment |
| 79% |
With potassium carbonate In acetone at 50℃; |
|
|
With potassium carbonate In ethanol for 2h; Reflux; |
|
Reference:
[1]Location in patent: scheme or table
Hall, Adrian; Billinton, Andy; Brown, Susan H.; Chowdhury, Anita; Clayton, Nicholas M.; Giblin, Gerard M.P.; Gibson, Mairi; Goldsmith, Paul A.; Hurst, David N.; Naylor, Alan; Peet, Caroline F.; Scoccitti, Tiziana; Wilson, Alexander W.; Winchester, Wendy
[Bioorganic and Medicinal Chemistry Letters, 2009, vol. 19, # 9, p. 2599 - 2603]
[2]Location in patent: experimental part
Whiting, Matthew; Harwood, Kathy; Hossner, Frank; Turner, Peter G.; Wilkinson, Mark C.
[Organic Process Research and Development, 2010, vol. 14, # 4, p. 820 - 831]
- 3
-
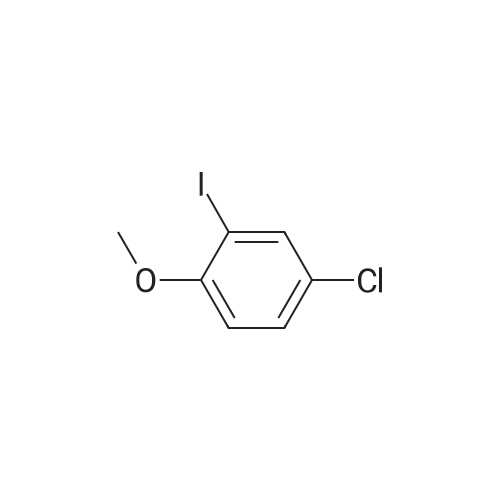 [ 52807-27-9 ]
[ 52807-27-9 ]

-
 [ 892663-09-1 ]
[ 892663-09-1 ]
- 4
-
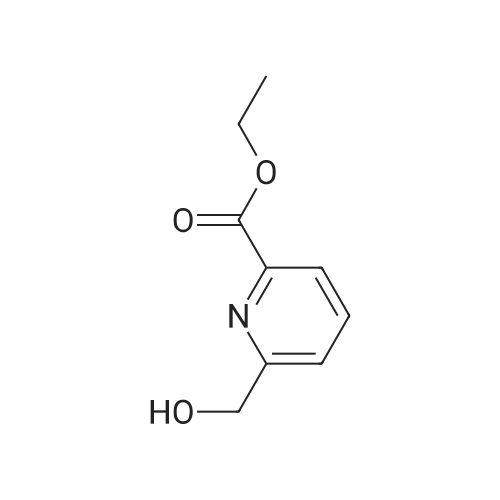 [ 41337-81-9 ]
[ 41337-81-9 ]

-
 [ 892663-09-1 ]
[ 892663-09-1 ]
| Yield | Reaction Conditions | Operation in experiment |
|
Multi-step reaction with 4 steps
1.1: sulfur trioxide pyridine complex; dimethyl sulfoxide; triethylamine / dichloromethane / 4 - 20 °C
2.1: ethylmagnesium bromide / dichloromethane; tert-butyl methyl ether / 0 - 23 °C
2.2: 2 h / 20 °C
3.1: palladium 10% on activated carbon; sulfuric acid; hydrogen; zinc dibromide / ethyl acetate / 6 h / 65 °C / 1520.1 Torr
4.1: potassium carbonate / ethanol / 2 h / Reflux |
|
|
Multi-step reaction with 2 steps
1: thionyl chloride / dichloromethane / 1 h / 10 - 25 °C
2: tris-(dibenzylideneacetone)dipalladium(0); potassium carbonate; DavePhos / ethanol; toluene / 3 h / 86 °C / Inert atmosphere; Large scale reaction |
|
|
Multi-step reaction with 4 steps
1.1: sulfur trioxide pyridine complex; dimethyl sulfoxide; triethylamine / dichloromethane / 4 - 20 °C
2.1: ethylmagnesium bromide / dichloromethane; tert-butyl methyl ether / 0 - 23 °C
2.2: 2 h / 20 °C
3.1: 10% palladium on activated charcoal; sulfuric acid; hydrogen; zinc(II) chloride / ethanol / 14 h / 50 °C
4.1: potassium carbonate / ethanol / 2 h / Reflux |
|
Reference:
[1]Whiting, Matthew; Harwood, Kathy; Hossner, Frank; Turner, Peter G.; Wilkinson, Mark C.
[Organic Process Research and Development, 2010, vol. 14, # 4, p. 820 - 831]
[2]Whiting, Matthew; Harwood, Kathy; Hossner, Frank; Turner, Peter G.; Wilkinson, Mark C.
[Organic Process Research and Development, 2010, vol. 14, # 4, p. 820 - 831]
[3]Whiting, Matthew; Harwood, Kathy; Hossner, Frank; Turner, Peter G.; Wilkinson, Mark C.
[Organic Process Research and Development, 2010, vol. 14, # 4, p. 820 - 831]
Categories

 Chemistry
Chemistry
 Pharmaceutical Intermediates
Pharmaceutical Intermediates
 Inhibitors/Agonists
Inhibitors/Agonists
 Material Science
Material Science














 For Research Only
For Research Only
 110K+ Compounds
110K+ Compounds
 Competitive Price
Competitive Price
 1-2 Day Shipping
1-2 Day Shipping







