Alternatived Products of [ 91028-13-6 ]
Product Details of [ 91028-13-6 ]
| CAS No. : | 91028-13-6 |
MDL No. : | MFCD27932211 |
| Formula : |
C12H14O2
|
Boiling Point : |
- |
| Linear Structure Formula : | - |
InChI Key : | - |
| M.W : |
190.24
|
Pubchem ID : | - |
| Synonyms : |
|
Safety of [ 91028-13-6 ]
| Signal Word: | |
Class: | |
| Precautionary Statements: | |
UN#: | |
| Hazard Statements: | |
Packing Group: | |
Application In Synthesis of [ 91028-13-6 ]
* All experimental methods are cited from the reference, please refer to the original source for details. We do not guarantee the accuracy of the content in the reference.
- Upstream synthesis route of [ 91028-13-6 ]
- Downstream synthetic route of [ 91028-13-6 ]
- 1
-
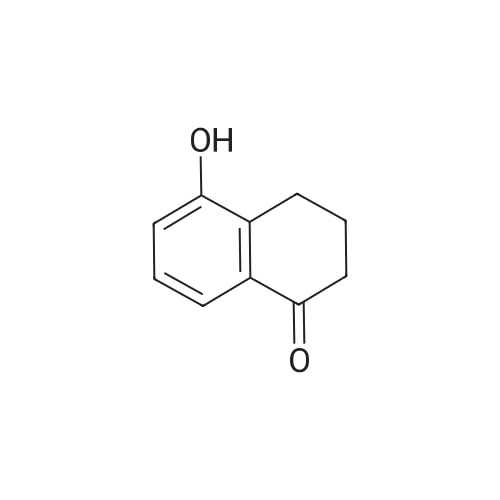
 [ 91028-13-6 ]
[ 91028-13-6 ]

-
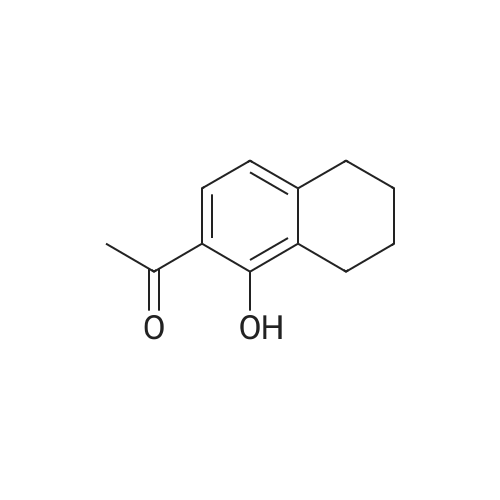 [ 95517-07-0 ]
[ 95517-07-0 ]
| Yield | Reaction Conditions | Operation in experiment |
| 100% |
With aluminium trichloride In 1,2-dichloro-benzene at 80℃; for 12h; |
|
|
With aluminium trichloride at 120℃; |
|
Reference:
[1]Pallavicini, Marco; Budriesi, Roberta; Fumagalli, Laura; Ioan, Pierfranco; Chiarini, Alberto; Bolchi, Cristiano; Ugenti, Maria Paola; Colleoni, Simona; Gobbi, Marco; Valoti, Ermanno
[Journal of Medicinal Chemistry, 2006, vol. 49, # 24, p. 7140 - 7149]
[2]Sergiewskaja; Morosowskaja
[Zhurnal Obshchei Khimii, 1944, vol. 14, p. 1107,1114][Chem.Abstr., 1946, p. 7186]
- 2
-
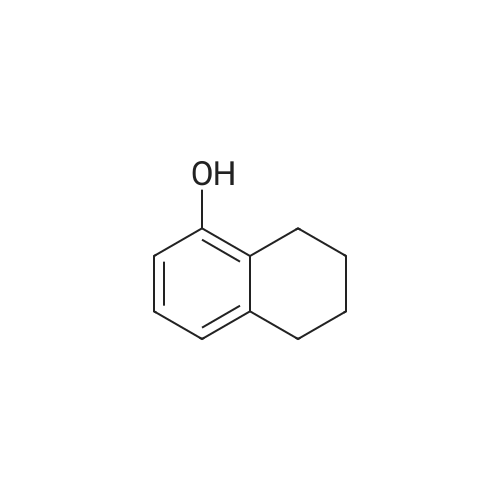 [ 529-35-1 ]
[ 529-35-1 ]

-
 [ 108-24-7 ]
[ 108-24-7 ]

-
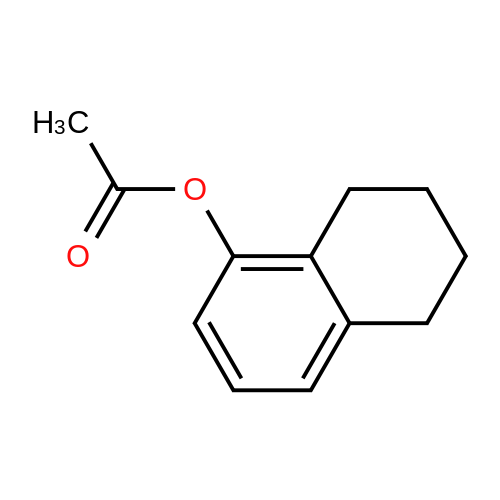
 [ 91028-13-6 ]
[ 91028-13-6 ]
| Yield | Reaction Conditions | Operation in experiment |
| 91% |
With triethylamine for 2h; Reflux; |
12.1
Step1: 5,6,7,8-tetrahydronaphthalen-1-ol (50.85 g, 340 mmol) was dissolved in acetic anhydride (500 ml) and triethylamine(56.8 ml, 408 mmol) was added to the reaction mixture. The whole was refluxed for 2h. The reaction mixture was cooled to room temperature and the solvent was removed under reduced pressure. The crude remaining liquid was dissolved into ethyl acetate (500mL) and the organic layer was washed several times with water and brine. The organic layer was then dried over Na2SO4, filtered, concentrated under reduced pressure to give a dark oil (65.4g; 91 % yield) LC: 4.94 min |
| 89% |
With pyridine; dmap at 20℃; Inert atmosphere; |
|
|
With sulfuric acid at 20℃; |
|
Reference:
[1]Current Patent Assignee: POXEL - EP2679591, 2014, A1
Location in patent: Paragraph 0082
[2]Guo, Weisi; Li, Ming; Li, Yufeng; Wang, Tao; Wen, Lirong; Zhang, Shanxue
[Organic Letters, 2022, vol. 24, # 8, p. 1742 - 1746]
[3]MOMOSE; OHKURA
[Pharmaceutical bulletin, 1956, vol. 4, # 3, p. 209 - 211]
- 3
-
 [ 529-35-1 ]
[ 529-35-1 ]

-
 [ 75-36-5 ]
[ 75-36-5 ]

-
 [ 91028-13-6 ]
[ 91028-13-6 ]
| Yield | Reaction Conditions | Operation in experiment |
| 100% |
With pyridine In dichloromethane for 3h; |
|
|
at 100℃; |
|
Reference:
[1]Pallavicini, Marco; Budriesi, Roberta; Fumagalli, Laura; Ioan, Pierfranco; Chiarini, Alberto; Bolchi, Cristiano; Ugenti, Maria Paola; Colleoni, Simona; Gobbi, Marco; Valoti, Ermanno
[Journal of Medicinal Chemistry, 2006, vol. 49, # 24, p. 7140 - 7149]
[2]Ssergijewsskaja; Morosowsskaja
[Zhurnal Obshchei Khimii, 1944, vol. 14, p. 1107,1114][Chem.Abstr., 1946, p. 7186]
Categories

 Chemistry
Chemistry
 Pharmaceutical Intermediates
Pharmaceutical Intermediates
 Inhibitors/Agonists
Inhibitors/Agonists
 Material Science
Material Science














 For Research Only
For Research Only
 110K+ Compounds
110K+ Compounds
 Competitive Price
Competitive Price
 1-2 Day Shipping
1-2 Day Shipping