| 97% |
With 1H-imidazole; dmap In N,N-dimethyl-formamide at 20℃; |
6.a
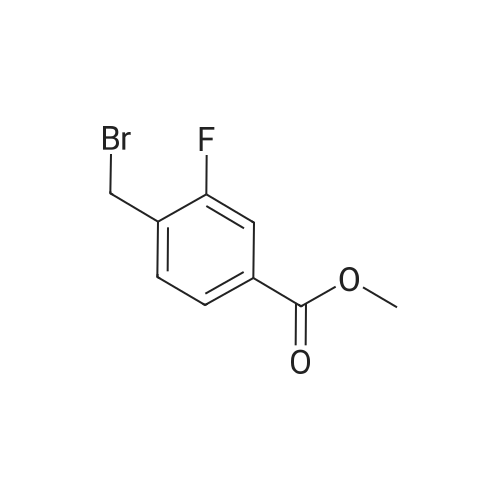
To 5.10 g (24.9 mmol) (4-bromo-2-fluoro-phenyl)-methanol in 30 mL DMF is added subsequently 4.06 g (26.1 mmol) fert-butyl-chloro-dimethyl silane, 2.57 g (37.3 mmol) imidazole and 456 mg (3.73 mmol) DMAP. The reaction mixture is stirred overnight at RT and the solvent is evaporated. The residue is diluted with EtOAc, washed five times with water and the combined organic phase is dried over MgSO4, filtered and the solvent is evaporated. Yield: 7.70 g (97% of theory); Rrvalue: 0.9 (silica gel, mixture C). |
| 75% |
With 1H-imidazole In dichloromethane at 0 - 25℃; |
(4-bromo-2-fluoro-phenyl)methoxy-tert-butyl-dimethyl-silane (SI-9)
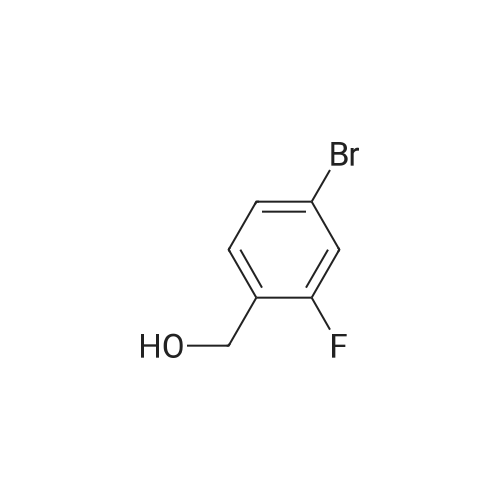
To a solution of compound SI-8 (12 g, 58.8 mmol) and imidazole (6.4 g, 94.1 mmol) inCH2Cl2 (200 mL) was added TBDMSCl (9.77 g, 64.8 mmol) at 0 oC over 1 hour and thenthe solution was stirred at room temperature overnight. Then, the reaction was quenchedwith water and extracted with EtOAc. The organic layer was washed with brine, dried over anhydrous Na2SO4, filtered and concentrated to give the crude product which was purified by column chromatography to obtain pure compound SI-9 (14 g, 75%) as a white solid. ESI-MS m/z 319 [M+H]+ calc. for C13H20BrFOSi. This intermediate was used in the next step without further characterization. |
| 70% |
With 1H-imidazole In dichloromethane at 20℃; for 1h; |
Synthesis of ((4-bromo-2-fluorobenzyl)oxy)(tert-butyl)dimethylsilane
Synthesis of ((4-bromo-2-fluorobenzyl)oxy)(tert-butyl)dimethylsilane A stirred solution of 4-bromo-2-fluoro benzaldehyde (49 g, 239 mmol, 1 eq) in DCM (400 mL) was treated with imidazole (32.5 g, 478 mmol, 2 eq) followed by TBDMS chloride (39.6 g, 263 mmol, 1.1 eq). The resulting solution was stirred at RT for 1 h, quenched with water (100 mL) and the layers were separated. The aqueous layer was extracted with DCM (2 χ 100 mL). The combined organic layer was washed with brine (100 mL), dried (Na2S04) and concentrated. The residue upon purification by column chromatography (silica gel 100-200, EtOAc/PE; 10:90) afforded ((4-bromo-2-fluorobenzyl)oxy)(tert- butyl)dimethylsilane (53 g, 70%) as a brown liquid. TLC system: EtOAc/ PE (1 :9), R,: 0.6 |
|
With 1H-imidazole In N,N-dimethyl-formamide at 20℃; for 10h; |
29.i
Example 29; 2-Butoxy-7,8-dihydro-9-[3-fluoro-4-{4-[3-(methoxycarbonylmethyl)phenoxymethyl]piperidin-1 -yl}methylbenzyl]-8-oxoadenine; [Show Image] Step (i); 4-Bromo-1-(tert-butyldimethylsiloxymethyl)-2-fluorobenzene; [Show Image] 4-Bromo-2-fluoro-1-(methoxycarbonyl)benzene 3.0g (12.9mmol) was dissolved in THF (50ml) and thereto was added portion wise under ice cooling lithium aluminum hydride 732mg (19.3mmol). After 1.5 hours, the reaction was quenched with 1N aqueous sodium hydroxide. After filtration over Celite, the filtrate was concentrated and thereto was added water. The mixture was extracted with ethyl acetate and the organic layer was washed with water and saturated brine, dried over Na2SO4, concentrated and dried in vacuo. The residue was dissolved in DMF (30ml) and thereto were added imidazole 871mg (12.8mmol) and TBSC1 1.93g (12.8mmol), followed by stirring at room temperature for 10 hours. After concentration, thereto was added water and the mixture was extracted with ethyl acetate. The organic layer was washed with water and saturated brine, dried over Na2SO4, and concentrated. The residue was purified by column chromatography to give the titled compound 2.7 g (8.5mmol) as a colorless transparent oil. Yield 69% 1H NMR (DMSO-d6) δ 7.35 (1H, dd, J = 7.7, 7.7 Hz), 7.27 (1H, dd, J = 1.8, 8.3 Hz), 7.16 (1H, dd, J = 1.8, 9.6 Hz), 4.72 (2H, s), 0.92 (9H, s), 0.10 (6H, s). |
|
With 1H-imidazole In N,N-dimethyl-formamide for 16h; |
2 Step 2: ((4-bromo-2-fluorobenzyl)oxy)(tert-butyl)dimethylsilane.
To a crude solution of 4-bromo-2-fluorophenyl)methanol (10.1 g, 49.3 mmol) in DMF (250 mL) was added TBSCl (11.1 g, 73.9 mmol) followed by Imidizole (6.71 g, 98.5 mmol). The solution was stirred for 16h. The crude solution was partitioned between EtOAc (500 mL) and LiCl (500 mL). The LiCl was discarded and the organic layer was washed with additional LiCl (2x250 mL). The combined organics were dried over Na2SO4 and concentrated to afford a crude oil which was purified via colomn chromatagrohy (Hexanes:EtOAc) to afford the title compound as a clear oil. |

 Chemistry
Chemistry
 Pharmaceutical Intermediates
Pharmaceutical Intermediates
 Inhibitors/Agonists
Inhibitors/Agonists
 Material Science
Material Science













 For Research Only
For Research Only
 110K+ Compounds
110K+ Compounds
 Competitive Price
Competitive Price
 1-2 Day Shipping
1-2 Day Shipping