Alternatived Products of [ 935447-39-5 ]
Product Details of [ 935447-39-5 ]
| CAS No. : | 935447-39-5 |
MDL No. : | MFCD11618051 |
| Formula : |
C6H4FNaO2S
|
Boiling Point : |
- |
| Linear Structure Formula : | - |
InChI Key : | - |
| M.W : |
182.15
|
Pubchem ID : | - |
| Synonyms : |
|
Safety of [ 935447-39-5 ]
| Signal Word: | |
Class: | |
| Precautionary Statements: | |
UN#: | |
| Hazard Statements: | |
Packing Group: | |
Application In Synthesis of [ 935447-39-5 ]
* All experimental methods are cited from the reference, please refer to the original source for details. We do not guarantee the accuracy of the content in the reference.
- Upstream synthesis route of [ 935447-39-5 ]
- Downstream synthetic route of [ 935447-39-5 ]
- 1
-
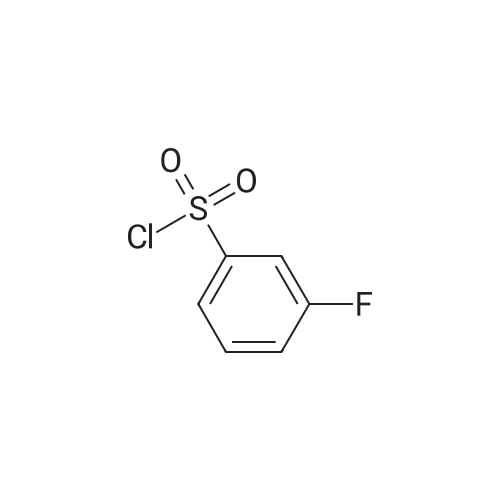 [ 701-27-9 ]
[ 701-27-9 ]

-
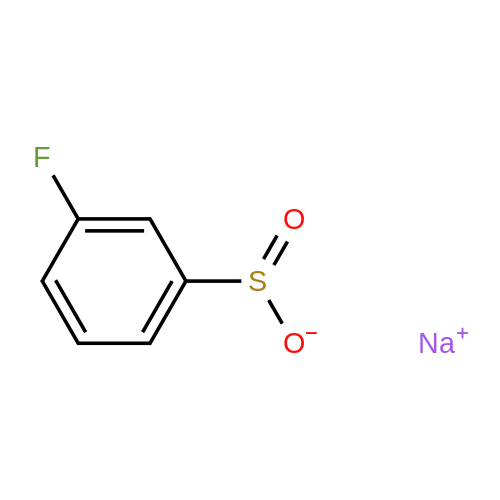 [ 935447-39-5 ]
[ 935447-39-5 ]
| Yield | Reaction Conditions | Operation in experiment |
| 98% |
With sodium carbonate; sodium sulfite; In water; at 50℃; for 2h; |
A solution of sodium sulfite (2.84 g, 22.5 mmol) in H20 (30 mL) was stirred at room temperature for 10 minutes. Base, such as sodium carbonate (3.18 g, 30.0 mmol), was added to the stirred solution. The resulting solution was stirred at elevated temperature, such as 50C for 10 minutes. 3-Fluorobenzenesulfonyl chloride 5.2 (2.0 mL, 15.0 mmol) was added dropwise to the solution and was stirred at 50C for 2 hours. The reaction mixture was evaporated to dryness and re-dissolved in EtOH (50 mL). The suspension was stirred at room temperature for 20 minutes. The suspension was filtered and the filtrate evaporated to afford a white solid, which was stirred with MeCN (20 mL) and then filtered to afford sodium 3-fluorobenzenesulfinate 6.2 (2.68 g, 98%) as a white solid. NMR (400 MHz, DMSO-d6) delta: 7.36 (m, 1 H), 7.29 (m, 1 H), 7.21 (m, 1 H) and 7.03 (m, 1 H). |
| 96% |
With sodium carbonate; sodium sulfite; In water; at 50℃; for 2h; |
General procedure: A solution of sodium sulfite (17.93g, 142.26 mmol) in H2O (100 ml) was stirred at room temperature for10 min. Sodium carbonate (23.90 g, 284.52 mmol)was added to the stirred solution.The resulting solution was stirred at 50 C for 10 minutes.cyclopropanesulfonyl chloride (20 g, 142.26 mmol) was added dropwise to thesolution and was stirred at 50 C for 2 hours. Thereaction mixture was evaporated to dryness and redissolved in EtOH (200 ml). The suspension was allowed to stir at roomtemperature for 20 min. The suspension was filtered and the filtrate evaporatedto afford a white solid, this was stirred with MeCN (50 ml) and thenfiltered to afford sodium cyclopropanesulfinicacid (13.70 g, 75 %) as a white solid. 1H NMR (400.132 MHz, DMSO d6)delta 1.38-1.31 (m, 1H), 0.27-0.24 (m,2H) and 0.08-0.03 (m, 2H). |
|
With sodium hydrogencarbonate; sodium sulfite; In water; at 80℃; for 3h; |
General procedure: The 4-cyanobenzenesulfonyl chloride (15 mmol, 1.0 equiv, 3.0 g) was dissolved in water (50 mL). Sodium sulfite (30 mmol, 2.0 equiv, 3.8 g) and sodium bicarbonate (30 mmol, 2.0 equiv, 2.5 g) were added, and the reaction mixture was reacted at 80 oC for 3 h. The solvent was evaporated and ethanol (150 mL) was added to the residue. The suspension was heated to 80 oC for 10 min, refluxed and filtered. The filtrate was evaporated, and then ethanol (100 mL) was added and heated to 80 oC for 10 min, refluxed and filtered at the second time. The solvent was evaporated under vacuum to give sodium 4-cyanobenzenesulfinate (1h, 1.99 g, 71%) as white powders. |
Reference:
[1]Patent: WO2018/65611,2018,A1 .Location in patent: Page/Page column 75; 89
[2]Tetrahedron Letters,2014,vol. 55,p. 3851 - 3855
[3]Molecules,2013,vol. 18,p. 97 - 113
[4]Green Chemistry,2015,vol. 17,p. 1395 - 1399
[5]Chemical Communications,2017,vol. 53,p. 2056 - 2059
[6]Tetrahedron Letters,2018,vol. 59,p. 4019 - 4023
- 2
-
 [ 935447-39-5 ]
[ 935447-39-5 ]

-
 [ 780-69-8 ]
[ 780-69-8 ]

-
 [ 2367-22-8 ]
[ 2367-22-8 ]
- 3
-
 [ 935447-39-5 ]
[ 935447-39-5 ]

-
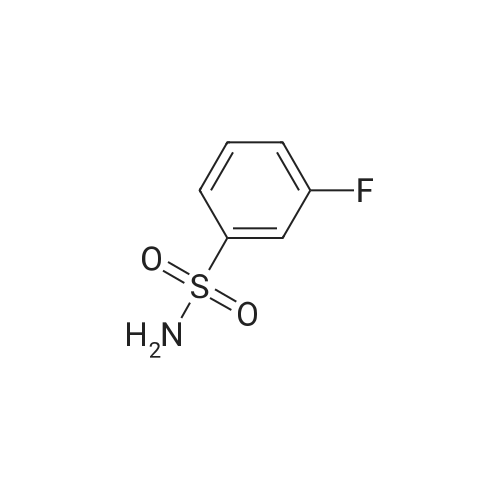 [ 1524-40-9 ]
[ 1524-40-9 ]
- 4
-
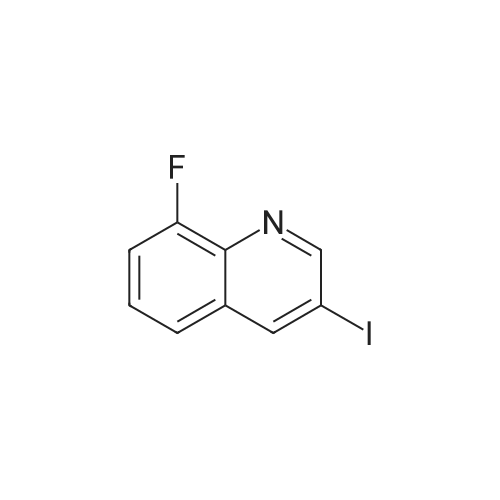 [ 866782-59-4 ]
[ 866782-59-4 ]

-
 [ 935447-39-5 ]
[ 935447-39-5 ]

-
8-fluoro-3-(3-fluorophenylsulfonyl)quinoline
[ No CAS ]
| Yield | Reaction Conditions | Operation in experiment |
| 29.7% |
With copper(I) trifluoromethanesulfonate benzene In N,N-dimethyl-formamide at 20 - 65℃; |
16 Example 16 1-[3-[[8-(4-Hydroxy-1-piperidyl)-3-quinolyl]sulfonyl]phenyl]piperidin-4-ol
Example 16 1-[3-[[8-(4-Hydroxy-1-piperidyl)-3-quinolyl]sulfonyl]phenyl]piperidin-4-ol (0347) 8-Fluoro-3-iodoquinoline (4 g, 14.65 mmol), sodium 3-fluorobenzenesulfinate (4.8 g, 26.4 mmol) and copper(I) trifluoromethanesulfonate benzene complex (8.19 g, 14.65 mmol) were suspended in DMF (80 ml) and warmed up to 65° C. for 4 h. The reaction mixture was stirred at room temperature over the weekend. The reaction mixture was filtered over a fritted funnel and concentrated. The residue was dissolved in DCM and washed several times with 2% aqueous NH3-solution and then several times with brine until neutral. The organic layer was dried with sodium sulfate, filtered and concentrated. The residue was purified using flash chromatography (120 g column; DCM 100%→DCM: MeOH 85:15). The crude material was purified using Chromabond flash chromatography to give 8-fluoro-3-(3-fluorophenylsulfonyl)quinoline (1.4 g, yield 29.7%). |

 Chemistry
Chemistry
 Pharmaceutical Intermediates
Pharmaceutical Intermediates
 Inhibitors/Agonists
Inhibitors/Agonists
 Material Science
Material Science















 For Research Only
For Research Only
 110K+ Compounds
110K+ Compounds
 Competitive Price
Competitive Price
 1-2 Day Shipping
1-2 Day Shipping








